Schizophrenia is a condition that, like many other neuropsychiatric conditions, is not well understood. Because it is not well understood, treating and developing new treatments for schizophrenia are difficult. One treatment mechanism lies in the Wnt signaling pathway. This pathway is mostly observed in nervous system development and neural circuit function. Because of this, some theorize that disruptions in prenatal and/or postnatal brain development could be triggers for schizophrenia.
Symptoms of Schizophrenia
:max_bytes(150000):strip_icc():format(webp)/schizophrenia-sign-symptoms-5095511_final-57a52853a10c4edcb4bd6a3b31f041ea.jpg)
The symptoms of schizophrenia cause it to be one of the leading causes of disability in the United States. According to VeryWellHealth.com, symptoms are categorized as positive, negative, or cognitive. Positive (also called psychotic) symptoms are added to an individuals perception of the world. Positive symptoms occur with delusions, hallucinations, disorganized thinking, or abnormal behavior such as agitation and involuntary or uncoordinated behavior. Negative symptoms occur with a reduction in typical behaviors which includes lack of motivation, interest in daily activities, and emotion, social withdrawal, and difficulty with typical functioning. Cognitive symptoms disrupt memory, attention, and concentration and can manifest as difficulty processing information and making decisions, inability to pay attention or concentrate, and difficulty using information that is heard [2].
Most of the treatments available for schizophrenia can help alleviate some of the positive symptoms of the disease but do not treat the underlying cause. A potential root cause treatment mechanism lies in the Wnt signaling pathway [3].
How Does Wnt Signaling Work?
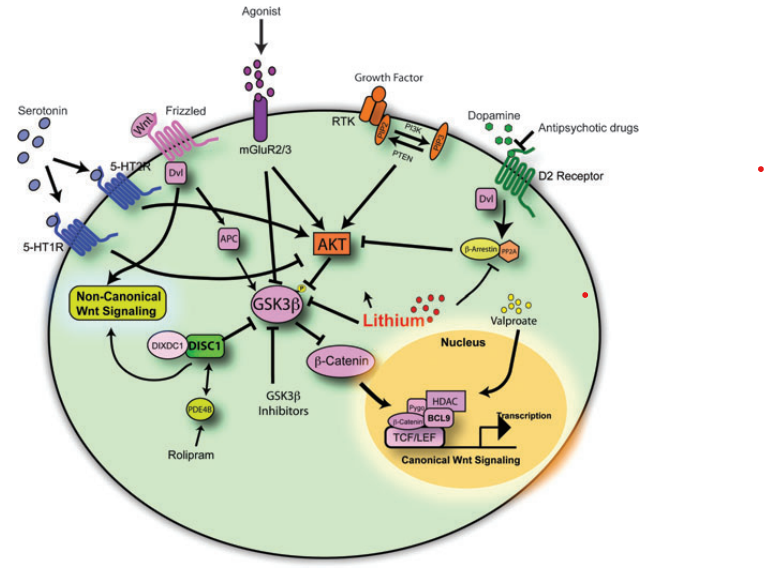
There are multiple Wnt pathways which have different functions. The canonical or traditional pathway is most related to the development and treatment of schizophrenia and will be discussed in further detail. Each of the Wnt pathways uses Wnt protein as a signaling ligand which begins an intracellular cascade of effects. When there is no ligand binding to the receptor in the canonical Wnt pathway, β-catenin (a transcription factor) is not phosphorylated and as a result is degraded by a GSK destruction complex. When β-catenin is destroyed, it cannot perform gene transcription in the nucleus. In summary, lack of Wnt signaling is correlated to increased GSK and decreased β-catenin [3].
When Wnt is available to bind do a receptor as a ligand, disheveled protein dissociates the GSK destruction complex. This decreases the phosphorylation of β-catenin which allows for it to have increased stability in the cytoplasm of the cell. The stable β-catenin is then transferred to the nucleus where it can initiate gene transcription. Canonical Wnt signaling allows for increased β-catenin levels and decreased GSK [3].
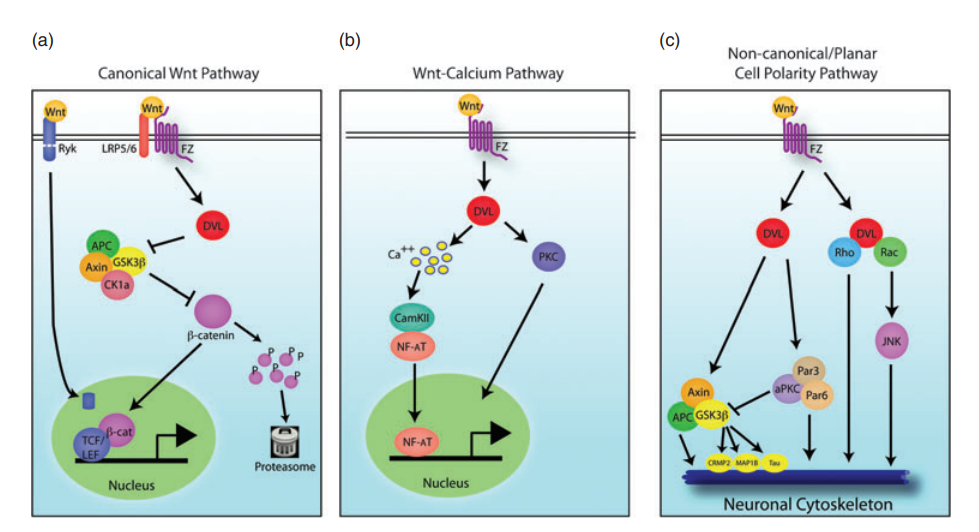
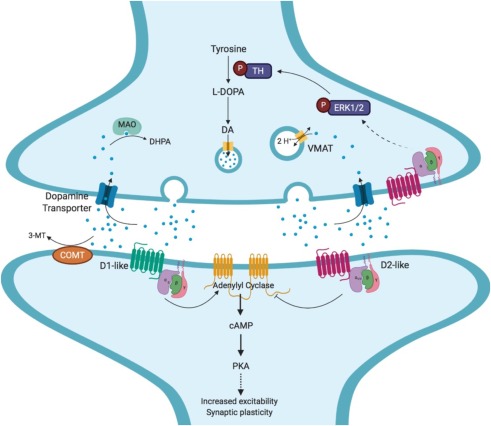
The non-canonical Wnt pathways can also affect neural circuit formation and synaptic plasticity in the central nervous system without the activity of β-catenin, as shown in Figure 1.
Neural Development
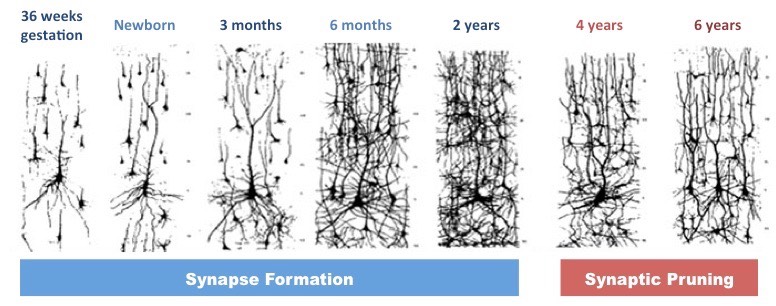
Wnt Dysfunction in Schizophrenia
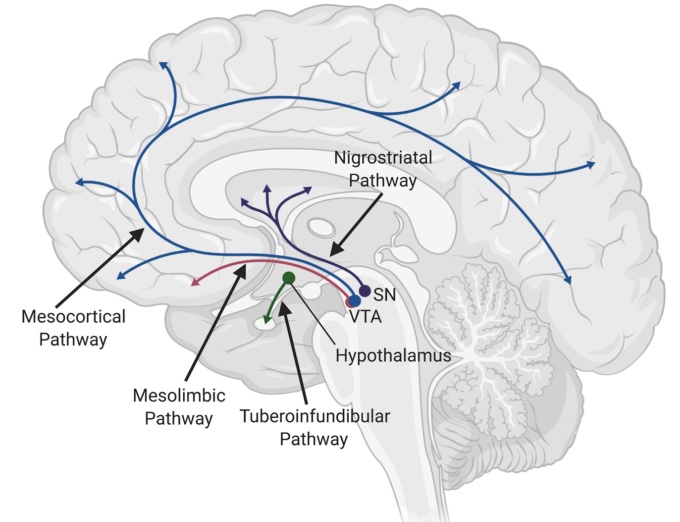
Individuals with Schizophrenia tend to have overactive dopamine receptors which oppose Wnt signaling. Dopamine will increase GSK dephosphorylation activation, which will phosphorylate β-catenin, increasing its degradation. In contrast to this, proper Wnt signaling would phosphorylate and inactivate GSK, thereby decreasing β-catenin phosphorylation, allowing for increased β-catenin concentration which acts to promote transcription. To further support this theory, several genetic variations associated with schizophrenia or other neurodevelopmental disorders have been associated with Wnt signaling [3].
Treatment Using the Wnt Pathway
Wnt signaling is a direct or indirect target of many drug treatments for schizophrenia or other psychiatric conditions. Antipsychotic treatments often inhibit D2 dopamine receptors which can decrease GSK activity, allowing the Wnt signal to be more active. Lithium is another commonly used neuropsychiatric treatment that can directly activate the Wnt pathway. By inhibiting GSK, lithium allows more β-catenin to accumulate and perform transcription functions. Metabotropic glutamate receptor (mGluR2/3) agonists can also be used to activate Akt which will phosphorylate/inhibit GSK3 which can result in increased disheveled protein and β-catenin. Each of these effects can be seen below in Figure 3 [3].






 P
P