How This Class Brought My Learning Together
As this semester ends, I have taken some time to think about how much I have grown not just in this class, but during my time at Concordia as a whole. This course helped me bring together so many skills and lessons I have picked up throughout college. I saw how everything I have learned connects in real life and how it’s helped shape me into someone who is more confident, open-minded, and ready to take on new challenges. Through this class, I feel like I have truly experienced what Concordia’s CORE curriculum and liberal arts education are all about learning that goes beyond just the classroom.
Learning to Enjoy Reading and Speaking Up
At the start of the semester, I did not feel very confident reading literature, especially when the language was hard or the ideas were complicated. I often felt nervous and unsure, like I didn’t really understand what I was reading. Honestly, I used to dread literature readings in class because they made me feel kind of dumb. But this class changed that. Since we had so many discussions, I learned it’s okay not to understand everything right away. Talking with classmates helped me see things more clearly, and I realized I wasn’t the only one who felt confused sometimes. Little by little, I stopped being afraid to ask questions or share my thoughts, even if I wasn’t completely sure. That gave me more confidence and helped me enjoy learning.
I now feel more comfortable reading challenging material, and I even enjoy it. That’s a big change for me and it ties into one of Concordia’s main goals: to instill a love for learning. I have started to see reading not as something I have to do, but something I get to do. I enjoy trying to figure out what the author means, and I like hearing different interpretations from my classmates. This back-and-forth helped me grow not just as a student, but as a thinker.
Getting Better at Research and Sharing Ideas
Another major skill I improved this semester is research. I used to think research was just about finding the right sources and putting together quotes. But in this class, I learned that good research is more than that it’s about asking deeper questions, connecting ideas, and making your own argument using what you have found. I also learned how to communicate my research better, both in writing and when talking to others. I had to think about how to explain things clearly, and that helped me better understand the material myself.
If I were updating my resume today, I would definitely add that I improved my research and communication skills this semester. Those are skills I know I will use in the future, especially because I am hoping to go to grad school for neuroscience which is mostly about research.
Learning from Others and Expanding My Perspective
One thing I really appreciated about this class was how much I learned from other people. Every week, we were encouraged to talk and share our thoughts, and that gave me the chance to hear so many different perspectives. Sometimes a classmate would say something about a reading that I had never even considered, and it completely changed how I saw it. Moments like that reminded me how important it is to really listen and stay open-minded, because there’s always more than one way to see something.
Even though we were mostly reading literature, our conversations went far beyond that. We ended up talking about real-world issues, history, culture, and even science. It made the material feel more connected to everyday life and helped me see how everything we learn is linked in some way.
Seeing Learning in a New Way
Before coming to Concordia, and even in my early years of college, I thought learning had to be a certain way. I used to think science classes were just about memorizing facts, and literature classes were only about writing essays. But being at a liberal arts school changed that for me. I started to see that learning can be more flexible and meaningful. For example, before I took neurochemistry, I expected it to be all lectures and exams. But it turned out to be more like a conversation where we could ask big questions and connect ideas from different subjects to understand things better.
This class especially helped me step back and see the bigger picture. Instead of just memorizing for a test, I learned to actually think about what I was learning and how it all fits together. That meant a lot to me.
What Liberal Arts Learning Means to Me
To me, learning at a liberal arts school like Concordia means getting a full picture of the world. It’s not just about preparing for a job, it’s about learning how to think, how to ask good questions, and how to be a better human being. We learn not just facts, but how to connect ideas, how to understand different cultures, and how to think about our values. This class reminded me that being educated means being able to listen, to speak up with respect, and to keep learning even outside of school.
Concordia talks a lot about BREW ( Becoming Responsibly Engaged in the World) and I feel like I understand what that really means now. It’s about using what I have learned to help others, to be thoughtful in my choices, and to stay involved in the world around me. Whether I’m working, studying, or just living in my community, I want to keep asking questions and growing.
A Real Example of Interdisciplinary Thinking
One example that really changed how I think is how I have come to understand autism. Before college, I mostly thought of it as a mental illness because that’s how it’s usually talked about in the media or in medical terms. But through this class and other experiences at Concordia, I started to realize there’s so much more to it.
In psychology and neuroscience, I learned about all the science and facts about how the brain works with autism. But what really opened my eyes were conversations around sociology and disability studies. I began to see that the biggest challenges autistic people face often come from how society treats them, not from autism itself. That made me rethink what it means to be “normal” and how we define ability in the first place.
I also started thinking about autism as something that’s not broken that needs to be fixed, but just a different way of thinking and experiencing the world. That shift felt important. It helped me see the value of being open to other ways of thinking, even when they are unfamiliar to me.
Conclusion: More Than a Class
This class was more than just reading articles or having discussions, it helped me grow as a person. It gave me the courage to speak up, to trust my thoughts, and to take chances, even when I felt unsure. I have realized that I’m not the only one who feels scared sometimes and that it’s okay. We’re all figuring things out. This course brought together everything I have been working on at Concordia: finding my voice, connecting ideas across subjects, and staying curious and open. As I move forward, I will carry the reminder that learning isn’t just about facts, it’s about understanding yourself, others, and the world a little better every day.





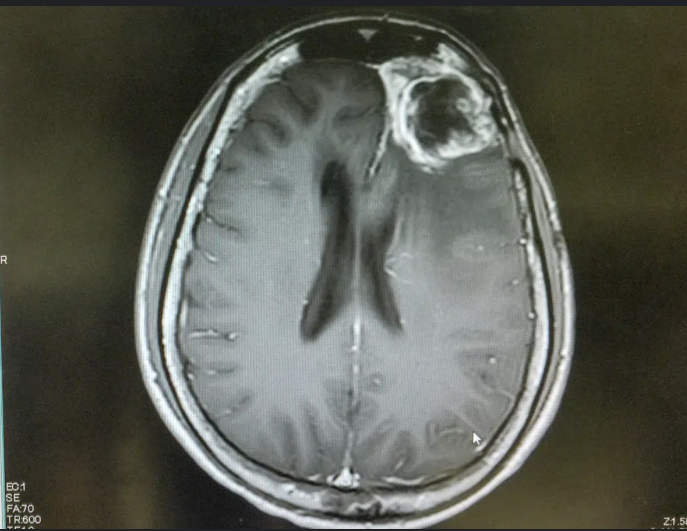
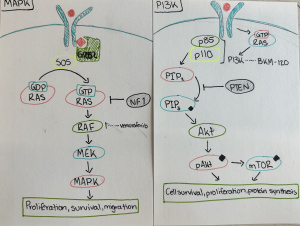
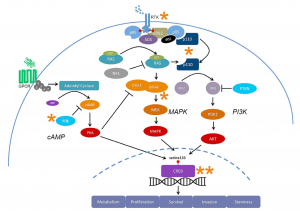





 Abstract by Alisha Debleye depicting the tumorous and dangerous growth in the human brain that can sometimes go untreated or undiagnosed.
Abstract by Alisha Debleye depicting the tumorous and dangerous growth in the human brain that can sometimes go untreated or undiagnosed.