Insulin – a hormone involved in more than just Diabetes
Type 2 Diabetes
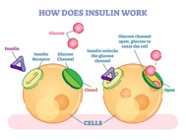
Type 2 diabetes is a chronic condition that affects how the body processes glucose. Insulin is the molecule that allows glucose to enter the body’s cells. The cell breaks down glucose to produce energy. In type 2 diabetes, the body cannot produce enough insulin, or it resists insulin. In both cases, the blood will be too concentrated with glucose, leading to hyperglycemia. This is the mechanism that produces type 2 diabetes, but is there another disease that has insulin resistance as a factor?
Alzheimer’s Disease
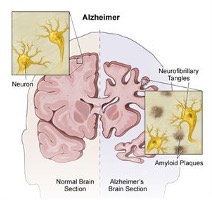
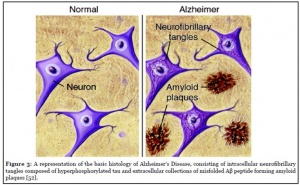
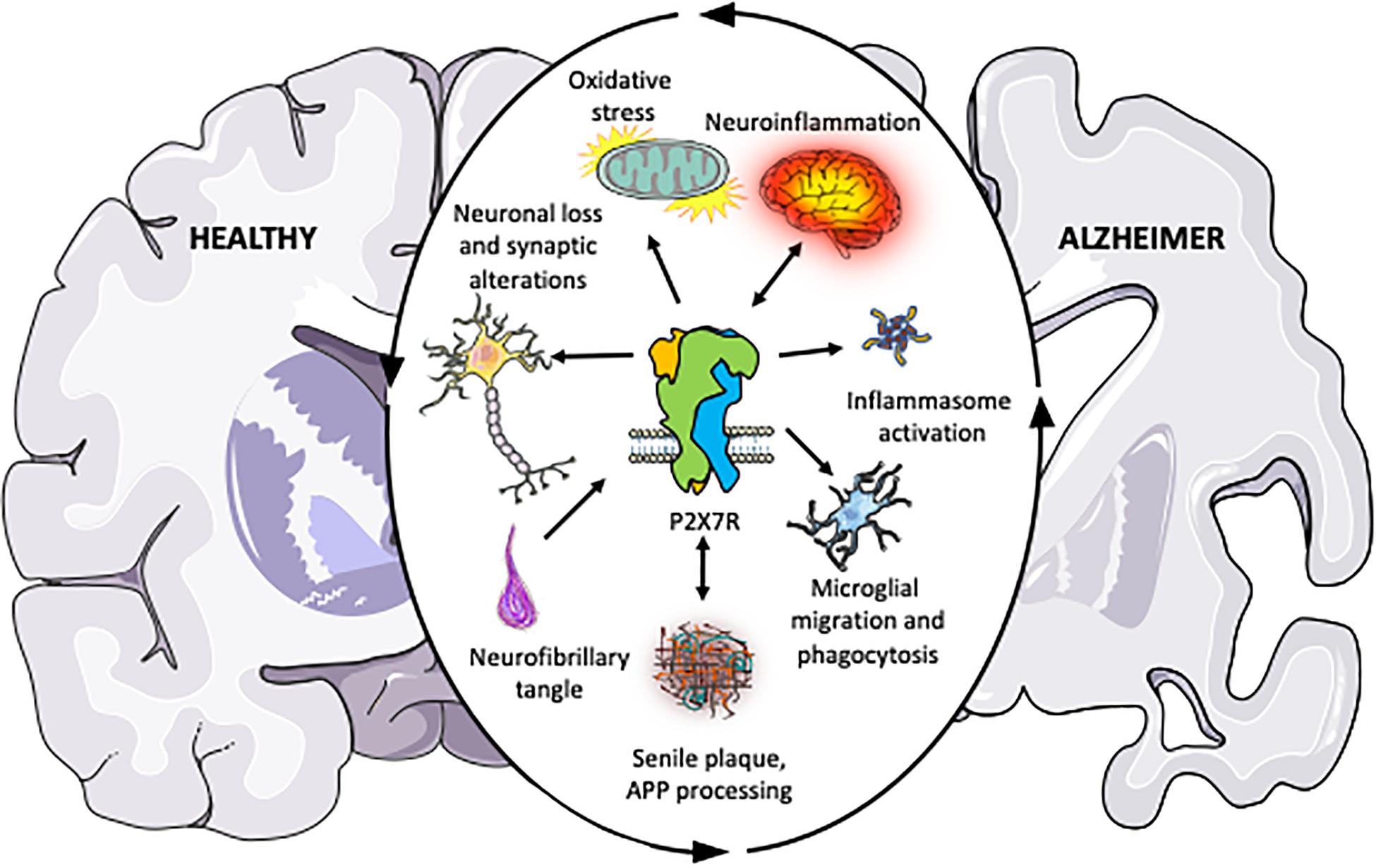
Alzheimer’s disease (AD) is the most common form of dementia, characterized as a progressive disease of memory loss, mood changes, disorientation, confusion, behavior changes, loss of physical functions, and an overall decrease of brain functioning. On the inside, neurofibrillary tangles, and amyloid -plaques are observed as key changes to the brain. Both of these phenomenon cause brain cells (neurons) to die.
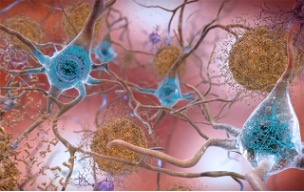
The neurofibrillary tangles are an abnormal accumulation of a protein, tau, that is hyperphosphorylated (too activated). Normally, tau supports the structure of the neuron by stabilizing microtubules which help guide nutrients and molecules down the cell. In AD, hyperphosphorylated tau detaches from the microtubules and stick to each other, forming tangles that block the neuron’s transport system from inside the cell.
Amyloid-beta plaques are clumps of beta-amyloid proteins that collect between neurons disrupting communication and inducing cell death. The accumulation of beta-amyloid proteins is in part due to an increase in the activation of the pathways that produce it. But how does tau become hyperphosphorylated and how do beta-amyloid pathways become activated? This is where insulin can be an answer.
Insulin in the Brain
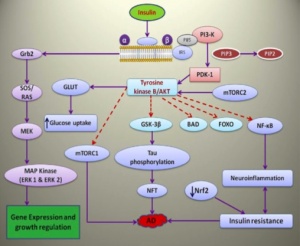
Insulin regulates glucose metabolism. The brain requires a constant abundance of glucose as it is the most active part of our body and, thus requires a great amount of energy. As seen above, insulin controls glucose channels to allow glucose uptake by the cell. However, in the brain, the glucose receptors that allow glucose intake into most brain cells are independent of insulin. They do not need insulin to open up. So why does the brain need insulin?
Insulin appears to be important for the regulation of food consumption behavior and monitoring energy stores. Insulin is a strong neuroprotective agent that acts against apoptosis, -amyloid toxicity, and oxidative stress. Insulin contributes to the control of nutrient homeostasis, reproduction, cognition, and memory.
But, what happens if the body becomes resistant to insulin like in Type 2 Diabetes?
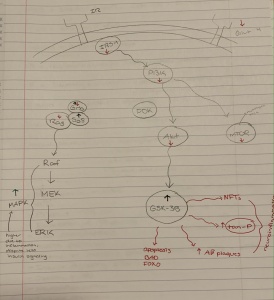
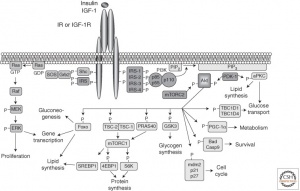
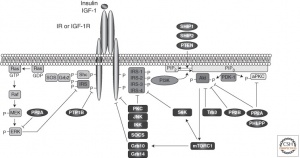
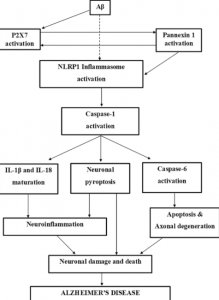
Insulin resistance (IR) induces the degradation of insulin-degrading enzyme (IDE), a key enzyme for clearing -amyloid proteins. IR activates the MAPK signaling pathway (an important brain pathway) which stimulates the amyloidogenic pathway and inhibits the non-amyloidogenic pathway leading to the accumulation of -amyloid. These malfunctions together lead to the formation of amyloid- plaques.
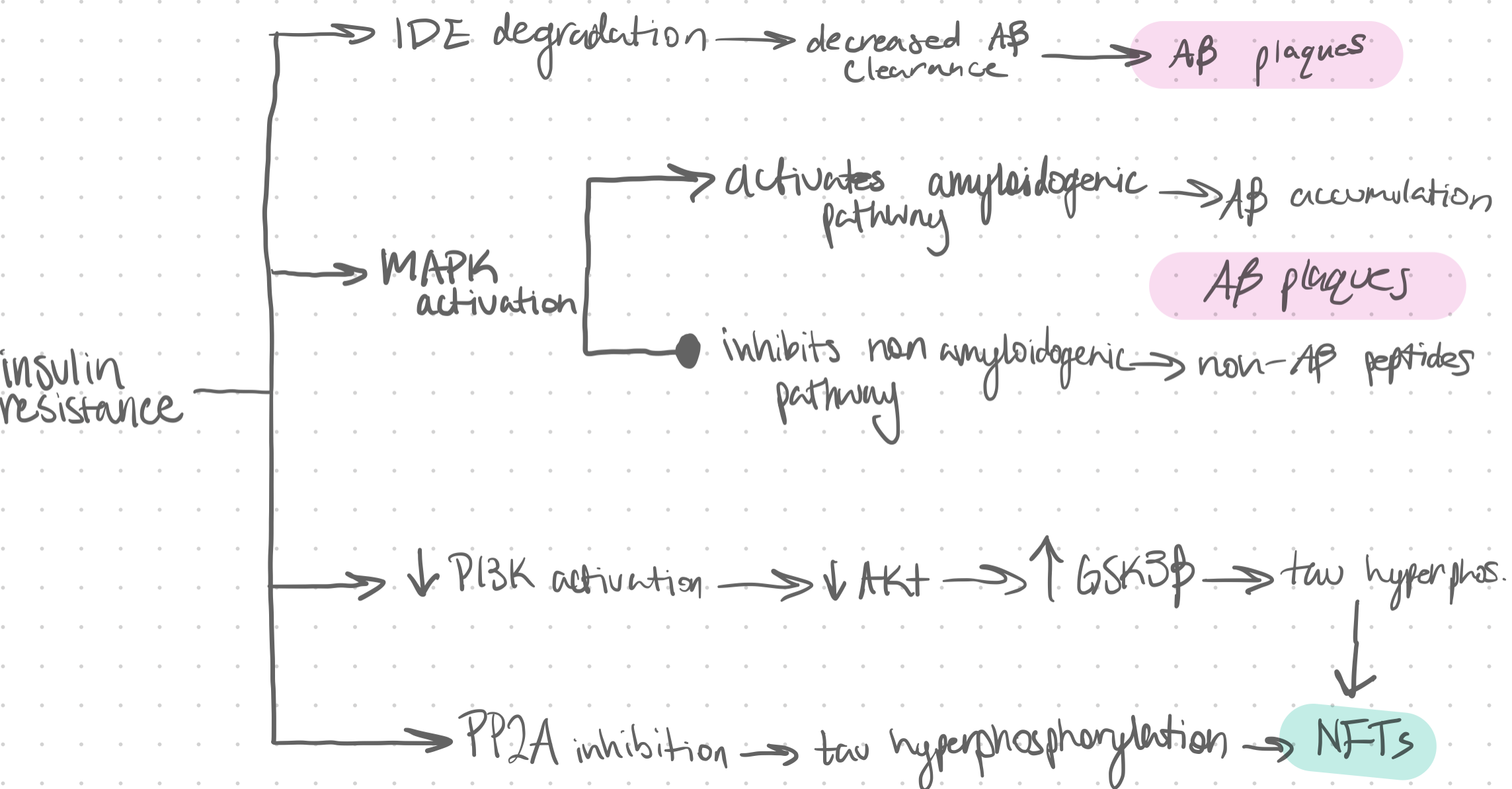
Additionally, IR decreases the activation of the PI3K/Akt pathway. Without this pathway being activated an enzyme called GSK3 is activated and phosphorylates tau. Another enzyme known as PP2A is inhibited by IR, which leads to tau phosphorylation. These two mechanisms combined instigate neurofibrillary tangles to form. A summary of these mechanisms is shown in figure 4.
Why should we care?
Alzheimer’s is a disease that affects millions of patients every year, and it’s not just the individuals with a diagnosis that are affected. Families, friends, nurses, doctors, administrators, and more are subjected to the hardships that come with AD.
If insulin resistance can be named as a risk factor for AD development, we can increase our arsenal of knowledge for how to decrease the severity of AD symptoms and push back the onset age.
But type 2 diabetes is linked to obesity. Obesity is concurrent to the overindulgence of unhealthy food and a low physical activity level. If obesity leads to insulin resistance, then obesity can lead to AD.
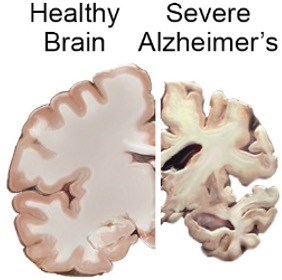
Therefore, eating right and maintaining a steady level of physical activity to avoid gaining weight can also lessen your chances of developing AD pathology and symptomology. So, the next time you reach for your fifth candy bar of the day or bail on your third walk of the week, think about how your brain could turn into this:
References:
https://doi.org/10.3389/fnhum.2020.602360
https://doi.org/10.1016/j.neuint.2020.104707
https://doi.org/10.1155/2015/105828