You may have heard that addiction is a disease, but this disease uniquely harms the body – it attacks learning.
Psychostimulant Use Disorder (PUD) is a chronic, relapsing condition characterized by compulsive drug-seeking behavior despite negative consequences[1]. Despite its prevalence, there are no FDA-approved medications specifically for treating PUD. Recent research, however, has pinpointed metabotropic glutamate receptors (mGluRs) as key players in the neuroplastic changes underlying addiction. “A review on the role of metabotropic glutamate receptors in neuroplasticity following psychostimulant use disorder”[2] delves into how these receptors modulate synaptic plasticity in brain reward circuits following psychostimulant use. This blog post will break down the science highlighted in the article and discuss possible therapeutic interventions for PUD.
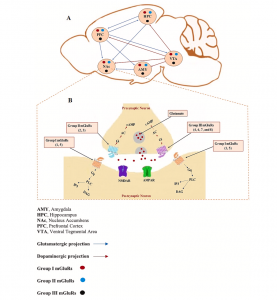
Addiction hijacks the brain’s reward system, which includes the ventral tegmental area (VTA), nucleus accumbens (NAc), prefrontal cortex (PFC), and amygdala. Psychostimulants like cocaine, amphetamines, and nicotine disrupt glutamate signaling (the brain’s primary excitatory neurotransmitter) leading to long-term synaptic changes[2]. These synaptic changes are extremely similar to those observed in learning. Instead of learning a new word in Spanish, a mathematics formula, or a fun psychology fact, the brain is learning that psychostimulant use feels good. These positive feelings are due to the drugs increasing extracellular dopamine and glutamate, reinforcing drug-associated memories and cravings. Glutamate acts through two receptor families: ionotropic receptors (fast-acting ligand-gated channels) critical for synaptic plasticity, and metabotropic receptors (mGluRs, which are G-protein coupled receptors) that modulate synaptic strength over longer time periods. The review focuses on mGluRs, divided into three groups based on signaling pathways and localization.
Group I mGluRs (mGluR1/5): Synaptic Potentiation and Relapse
Group I mGluRs are located postsynaptically and are linked to Gq proteins. They activate phospholipase C (PLC) and PKC, which hydrolyze PIP2 into DAG and IP3. These intracellular events create various outcomes depending on the psychostimulant.
- Role in PUD:
- Cocaine: Enhances long-term potentiation in the NAc and PFC. Antagonists reduce drug-seeking, while agonists promote relapse.
- Amphetamines: Increase mGluR5 expression in the PFC, correlating with conditioned place preference.
- Nicotine: mGluR5 blockade via antagonists attenuates nicotine self-administration and reward[2].
Group II and III mGluRs (mGluR2/3/4/6/7/8): Presynaptic Inhibition
Group II and III mGluRs are located presynaptically. They are Gi/o-coupled, which function to inhibit glutamate release (dampening the effects of the drugs) by decreasing cAMP and adenylyl cyclase levels. As with Group I, the intracellular inhibition creates varying effects depending on the type of drug.
- Role in PUD:
- Cocaine: Agonists inhibit relapse by normalizing hyper-glutamatergic states in the NAc. mGluR7 activation inhibits relapse by reducing glutamate release in the NAc.
- Methamphetamine: Downregulation of mGluR2/3 contributes to cravings, and agonists reduce reinstatement. mGluR8 expression changes may counteract drug-induced plasticity.
- Nicotine: Activation in the VTA decreases nicotine-seeking behavior[2].
Just as we are able to learn, we are able to unlearn. Viewing addiction as a problem with learning, association, and rewards provides possible promising therapeutic implications for something that is currently untreatable by medication. Targeting mGluRs offers a nuanced approach, and the article notes that Group I antagonists may prevent cue-induced cravings, Group II agonists could restore glutamate homeostasis, and Group III modulation might disrupt drug-associated memories[2]. Taken together, targeting mGluRs may have promising therapeutic benefits.
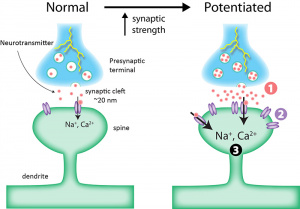
The review highlights mGluRs as critical mediators of addiction-related neuroplasticity. While recent research is promising, translating these findings into treatments requires further research, especially given the complexity of glutamate signaling across brain regions and its importance in a variety of normal brain functions. For now, mGluRs remain a hopeful future direction in curing PUD.
Addiction rewires the brain and creates a learned dependence on and craving for drugs, but by hacking glutamate’s mGluRs, researchers may just reset the system. Therefore, researching the therapeutic effects of mGluR agonists and antagonists is essential for combating PUD.
References
