Schizophrenia is one of the hardest mental conditions to treat and it can often be difficult to get patients to comply with a medication regiment. This is due to a relatively simple reason, that reason being that antipsychotics can make those who take it feel absolutely miserable. This is something that is well known by pharmaceutical companies and this has resulted in different generations of antipsychotics that each have their pros and cons. But to understand how these medications work we must understand how schizophrenia works.
How Schizophrenia Works
Schizophrenia has long been considered an issue with dopamine (DA), and this can be explained by the symptoms. The symptoms of schizophrenia have been associated with D2 receptor misfiring in the mesolimbic pathway leading to hallucinations, delusions and disorganized thinking. This has been supported through studies that show increasing DA release can induce psychosis and blocking DA relieves it. More recently, the hypothesis has changed from too much DA to DA dysregulation, with too much DA in some areas and not enough in others (1). This change in philosophy can also be seen with the mechanisms of third generation antipsychotics, which will be explained further on in this post. For more details on the DA hypothesis, check out this article.
Researchers have recently suggested schizophrenia is resulted from dysregulated Wnt signaling caused by DA misfiring. The way Wnt signaling works is relatively simple, and one must start at the destruction complex to understand it. When Wnt signaling is not active, there is a destruction complex that leads to the destruction of B-catenin. B-catenin is a transcription factor that is associated with cell/neuron growth. One of the most important parts of this complex is called GSK3B, this is basically what marks B-catenin for destruction. When Wnt signaling is active, it leads to the disassociation of the destruction complex leading to B-catenin being able to enter the nucleus. The theory behind all this is too much DA signaling during development leads to Wnt dysregulation and the lack 0f B-catenin getting into the nucleus results in abnormal brain pathways associated with schizophrenia. Now you are probably wondering what DA’s role in this is, rather than DA itself it relates more to what DA receptor activation leads to. First we must talk about something called AKT, when this becomes activated it inhibits GSK3B therefore leading to more B-catenin accumulation. D2 receptor activation leads to the inhibition of AKT and therefore strengthens the destruction complex (2). This has been supported in animal models exploring the role of GSK3B and AKT in schizophrenia and how it relates to DA and it can also be supported when looking at the way some antipsychotics work. For more details on this hypothesis of schizophrenia check out this article.
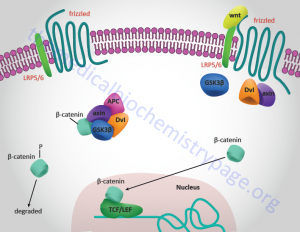
This is a visual on Wnt signaling to help with understanding. With inactive signaling on the left, and active signaling on the right. Sourced from.
First Generation Antipsychotics (3)
First generation antipsychotics (FGA) were first offered in 1952. These can result in some very unpleasant symptoms such as sedation and extrapyramidal symptoms, this means symptoms similar to those of Parkinson’s such as movement issues like dyskinesia or tremors. There are two kinds of FGAs, high potency, which have lower sedative symptoms but higher extrapyramidal symptoms, and low potency, which result in higher sedation and lower extrapyramidal symptoms.
While the mechanisms of action vary between different drugs, the common factor is blocking of D2 receptors in the substantia nigra and the mesolimbic pathway. As discussed previously, reduced D2 signaling results in more Wnt signaling. Additionally, many FGAs directly interact with Wnt signaling. Clozapine induces phosphorylation of GSK-3B and AKT, both of these result in the disassociation of the destruction complex and therefore more B-catenin can accumulate. Lithium, another FGA, prevents inactivation of AKT and also directly inhibits GSK3B, which also leads to more B-catenin accumulation.
The side effects of FGAs are the most severe out of all of the antipsychotic generations. The most prevalent of these side effects are the extrapyramidal effects. These result from blocking of D2 receptors in the substantia nigra, an area very important for motor planning and execution. This area is affected by Parkinson’s, which is why we see similar symptoms between the two. Disruption of the mesolimbic pathway can result in social apathy and social withdrawal, turning the patient into a shell of themselves. Additionally, FGAs interact with histamine receptors, which induces sedation. These side effects are seen in some form in most people who take FGAs, and therefore further development of drugs with less side effects was needed.
Second Generation Antipsychotics (4)
Second generation antipsychotics (SGAs) were first offered in 1990 and have been found to result in less severe side effects. With SGAs D2 receptors are still blocked in the mesolimbic pathway, but not as much in the substantia nigra as in FGAs. This results in less of the extrapyramidal symptoms seen in FGAs. Additionally, SGAs blocks serotonin receptors, primarily in the frontal cortex. This leads to more DA release which helps relieve some of the side effects caused by antipsychotics. SGAs have been found to increase Wnt signaling through D2 receptor blocking, similarly to FGAs. There is less evidence showing they are direct modulators similarly to FGAs like lithium, but through D2 receptors blocking, inherently increasing Wnt signaling.
Side effects of SGAs are similar to that of FGAs. The difference is seen in the decrease of extrapyramidal side effects. Due to less D2 blocking in the substantia nigra, less extrapyramidal side effects are seen in SGAs. There is still a risk of these side effects, but the risk is lower and the symptoms are also less severe than FGAs. SGAs also mess with histamine receptors, leading to sedation. Something unique to SGAs is they have been associated with weight gain and insulin resistance/type 2 diabetes. This is caused through serotonin receptor blockage, messing with the appetite and eating habits. While these side effects are a great improvement from FGAs, they are far from ideal. This has resulted in the development of more unique antipsychotics.
Third Generation Antipsychotics (5)
The mechanisms behind third generation antipsychotics (TGA) vary greatly, therefore, we will focus primarily on the most well studied one, aripiprazole. Aripiprazole was first offered in 2002 and does not work like other antipsychotics. Rather than blocking D2 receptors, it works by selectively firing them. What this means is that when there is too much DA, aripiprazole competes with it and binds to the receptor instead, acting as an antagonist in this context. When there is too little DA, aripiprazole picks up the slack, acting as an agonist in this context.
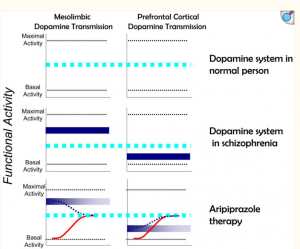
(Graph showing how aripiprazole acts depending on the context, source)
This functional selection of firing results in less side effects, and less severe side effects. The side effects are more or less the exact same just less risk of experiencing them and less severity. There are a few exceptions though. Something unique to TGAs is they actually can cause restlessness and insomnia rather than sedation due to continued firing of D2 receptors and less histamine receptor interaction.
The Future
As antipsychotics continue to be developed, schizophrenia treatment will continue to change and mold depending on the current literature. Antipsychotics have been around for 74 years, which in the grand scheme of things is very recently. There are people who developed schizophrenia before the discovery of antipsychotics still alive today, and while this fact can be depressing in a way, it can also be promising. They have come very far compared to how they started, and as treatment philosophy has started to shift from complete D2 receptor blocking to selective firing, the side effects will hopefully continue to improve.

(Source)
Feature image developed through Google Gemini 3 and Eli Hunt. Prompt “make an image that represents schizophrenia and the different generations of antipsychotics” followed by “make it more artistic, less words”
-
Brisch, R., Saniotis, A., Wolf, R., Bielau, H., Bernstein, H. G., Steiner, J., Bogerts, B., Braun, K., Jankowski, Z., Kumaratilake, J., Henneberg, M., & Gos, T. (2014). The role of dopamine in schizophrenia from a neurobiological and evolutionary perspective: old fashioned, but still in vogue. Frontiers in psychiatry, 5, 47. https://doi.org/10.3389/fpsyt.2014.00047
- Singh K. K. (2013). An emerging role for Wnt and GSK3 signaling pathways in schizophrenia. Clinical genetics, 83(6), 511–517. https://doi.org/10.1111/cge.12111
- Grace, A. A., & Uliana, D. L. (2023). Insights into the Mechanism of Action of Antipsychotic Drugs Derived from Animal Models: Standard of Care versus Novel Targets. International journal of molecular sciences, 24(15), 12374. https://doi.org/10.3390/ijms241512374
- Chokhawala, K. P., & Stevens, L. (2023). Antipsychotic Medications. In StatPearls. StatPearls Publishing.
- Mailman, R. B., & Murthy, V. (2010). Third generation antipsychotic drugs: partial agonism or receptor functional selectivity?. Current pharmaceutical design, 16(5), 488–501. https://doi.org/10.2174/138161210790361461