Schizophrenia affects about 1% of the global population,[1] and is a chronic psychiatric disorder that significantly contributes to disability worldwide. It is usually diagnosed in late adolescence and continues into adulthood. Current treatments focus on symptoms like hallucinations but don’t address the neurodevelopmental causes. And it’s increasingly recognized as a disorder of brain development, with early factors like maternal infections during pregnancy disrupting brain development and raising the risk of schizophrenia [2].
But despite this, the molecular mechanisms linking genetic and environmental factors are still unclear. While the canonical Wnt/β-catenin pathway has been a major focus, its role remains complex. Therefore, Recent evidence points to the role of Wnt signaling [3] and glycogen synthase kinase 3 (GSK3) pathways in schizophrenia, with non-canonical Wnt pathways like PCP and Wnt/calcium now playing key roles in shaping brain circuits and synaptic function in schizophrenia.
Cracking the Code of Brain Development: Meet the Non-Canonical Wnt Pathways ✨
Our brains are like a busy traffic system, where cars (cells) need to move to the right places and follow the correct paths to keep everything running smoothly. The non-canonical Wnt pathways, Wnt/PCP (Planar Cell Polarity) and Wnt/Ca²⁺ (Calcium) act like traffic signals, guiding the cells to their proper destinations and making sure they send the right signals. When these signals get messed up, like traffic lights malfunctioning, things can go wrong, leading to problems with brain development and function, as seen in schizophrenia.
But when these pathways get messed up, especially in conditions like schizophrenia, the dance falls apart. Neurons end up in the wrong places, signals get mixed up, and inflammation increases, which leads to problems in brain development and thinking.
Therefore, understanding these pathways better could help us figure out how schizophrenia develops and maybe even lead to better treatments.
Animal Models and Wnt Signaling in Schizophrenia
To understand how these pathways are involved in schizophrenia, animal models, particularly mice, are used in research. Mice do not have the exact same symptoms as humans, but they help researchers understand how brain pathways can go wrong in schizophrenia.
According to the paper “An emerging role for Wnt and GSK3 signaling pathways in schizophrenia,” a study with Dvl1 knockout (KO) mice (mice missing a key protein) showed issues with social behavior and prepulse inhibition (PPI) [4], which are similar to symptoms seen in schizophrenia patients. Since Dvl1 works in both canonical and non-canonical Wnt pathways, scientists believe that both pathways might contribute to the disorder. These findings highlight the involvement of both pathways in shaping behavior and brain function.[1]
What Are the Non-Canonical Wnt Pathways? (No β-Catenin Here!)
So, now that we’ve seen how these pathways influence schizophrenia, let’s take a deeper dive into what the non-canonical Wnt pathways actually are, and why they’re so important for brain health.
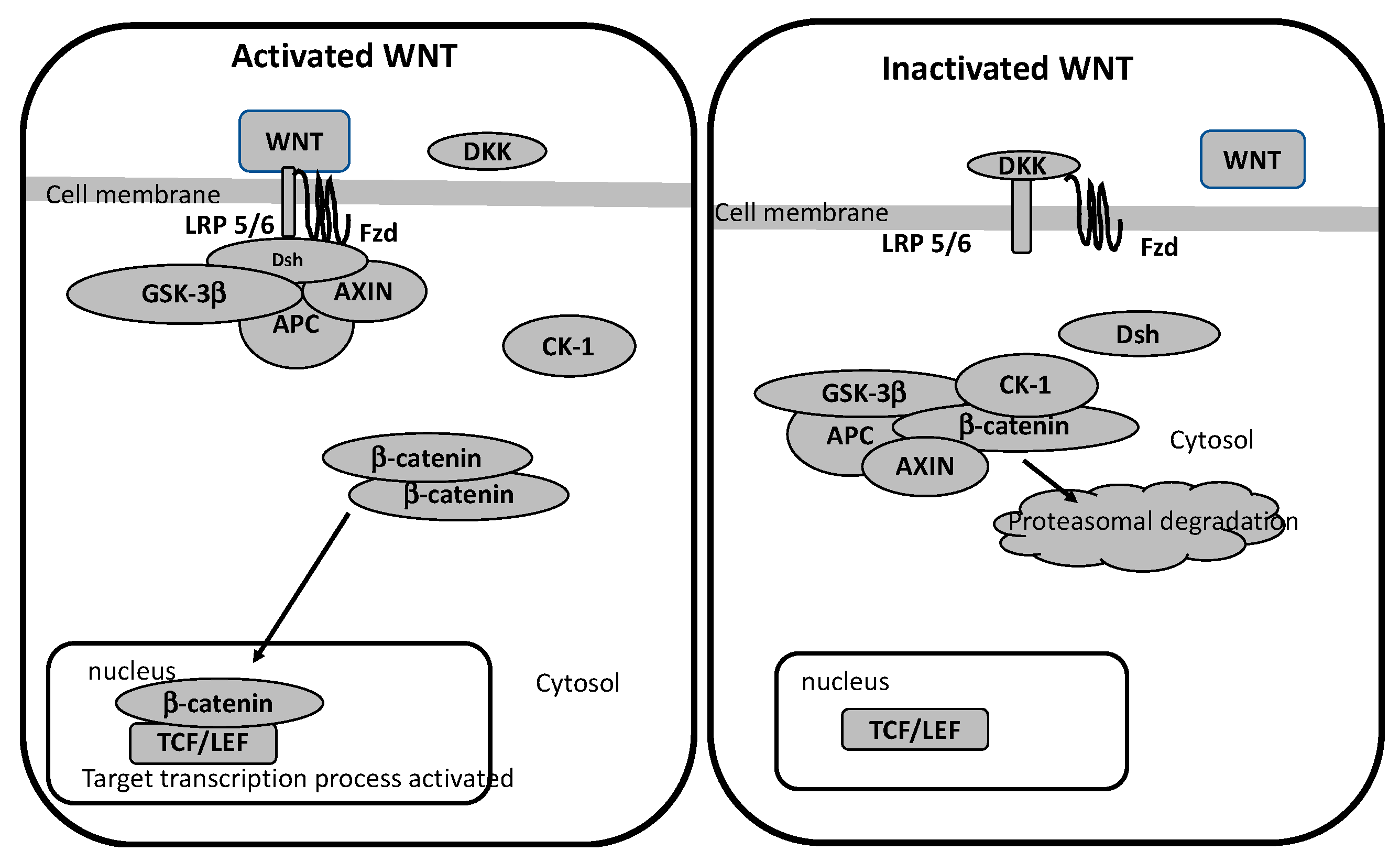
When most people think of Wnt pathways, they imagine the one that uses β-catenin. But the non-canonical pathways take a different route skipping β-catenin entirely. Instead, they focus on regulating cell movement, shape, and signaling key factors in ensuring that the brain develops correctly and functions smoothly. These pathways are like the hidden helpers of brain development, making sure everything stays in its right place.
1. Wnt/PCP (Planar Cell Polarity) Pathway
The Wnt/PCP pathway is like a GPS for cells. It tells them where to go and how to line up properly as the brain develops.
Key Players:
- Wnt Ligand: The signal that starts the process.
- Frizzled (FZD) Receptor: The docking station where Wnt binds.
- Coreceptors (ROR/RYK): Help guide the signal.
- Disheveled (DVL): A protein that wakes up and tells the cell to move.
- RhoA & Rac1: Proteins that change the cell’s shape and help it move.
How It Works:
- Wnt binds to Frizzled and ROR/RYK.
- Disheveled gets activated and turns on RhoA and Rac1.
- RhoA: Controls how rigid the cell is.
- Rac1: Helps the cell stretch and move.
❓ What Happens When It’s Disrupted in Schizophrenia?
When the Wnt/PCP pathway goes off track, it messes up the system that guides neurons to the right places in the brain. Here’s what happens when things go wrong:
- Defective Neuronal Migration: The PCP pathway helps neurons find their spot in the brain. If disrupted, neurons can end up in the wrong places, leading to faulty brain circuits and impaired information processing.
- Loss of Synaptic Organization: The pathway also helps cells line up properly. If it’s off, synapses get misaligned, making it harder for neurons to connect, affecting the brain’s ability to adapt and refine connections.
- Cytoskeleton Dysfunction: RhoA and Rac1 control the cell’s shape and movement. Disrupting the PCP pathway messes with these proteins, causing irregular cell shapes and making brain development even messier.[5]
These errors in cell migration, synapse alignment, and cytoskeleton organization could be the root cause of the structural abnormalities and cognitive deficits seen in schizophrenia
2. Wnt/Ca²⁺ (Calcium) Pathway ⚡
This pathway controls calcium signals inside the cell, which are super important for learning, memory, and controlling inflammation.
Key Players:
- Wnt Ligand: The signal that kicks things off.
- Frizzled (FZD) Receptor: The receptor that catches the Wnt signal.
- Phospholipase C (PLC): Splits a molecule called PIP2 to release calcium.
- Calcium (Ca²⁺): Sends signals inside the cell.
- CaMKII & PKC: Proteins that control synapses and inflammation.
How It Works:
- Wnt binds to Frizzled, activating PLC.
- PLC splits PIP2, which releases calcium inside the cell.
- The calcium sudden increase activates proteins that control synaptic strength and inflammation.

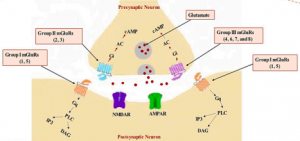
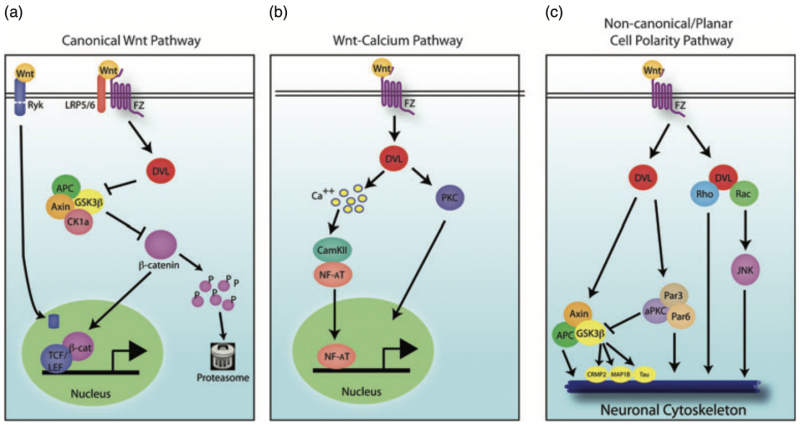
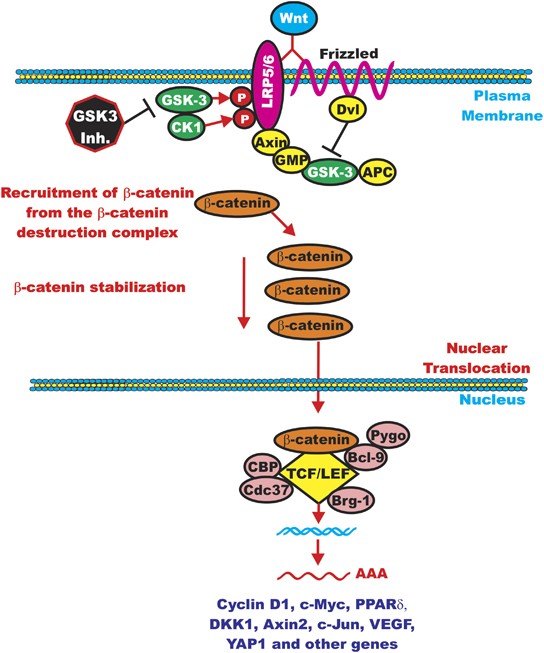
Figure 1: Shows a comparison between the canonical and non-canonical Wnt signaling pathways. (A) The canonical β-catenin-dependent pathway stabilizes β-catenin, allowing it to enter the nucleus and activate target genes. (B) The non-canonical pathways include the JNK/PCP pathway, which controls cytoskeletal organization, and the Ca²⁺ pathway, which regulates gene expression through calcium-dependent signaling. [6]
❓ What Happens When It’s Disrupted in Schizophrenia?
When this pathway is disrupted, calcium signaling becomes unregulated, leading to a buildup of intracellular calcium (Ca²⁺). This overload can trigger:
- Excessive activation of CaMKII and PKC: These proteins are important for how synapses strengthen and adapt, but too much activity can cause synaptic dysfunction, making it harder for neurons to communicate properly. [7]
- Increased neuroinflammation: Uncontrolled calcium can activate inflammatory responses, which release cytokines and other inflammatory molecules. Over time, this chronic inflammation can damage neurons and disrupt normal brain function.
- Oxidative stress and mitochondrial dysfunction: High levels of calcium can stress mitochondria, leading to oxidative damage, which further disrupts neuronal health.
This imbalance in calcium signaling could be one of the major reason for the synaptic dysfunction, cognitive decline, and increased inflammation often seen in schizophrenia.

Figure 2: Shows brain scans comparing inflammation in healthy individuals, high-risk individuals, and those with schizophrenia. The colored images indicate inflammation levels, with warmer colors (orange/yellow) showing higher inflammation. Inflammation increases from healthy to high-risk and is highest in individuals with schizophrenia. [8]
Conclusion: A Bright Future for Schizophrenia Treatment
Understanding how the brain develops and how disruptions in pathways like the non-canonical Wnt pathways contribute to schizophrenia is a big step forward.
The key takeaway? More research is needed! The better we understand these pathways, the more likely we are to develop treatments that focus on the cause of schizophrenia, not just its symptoms. With more knowledge, we can make real changes in the lives of those affected.
The brain is an amazing system, and with the right science, we can help restore balance, giving people with schizophrenia a brighter, healthier future. Stay hopeful science is making great progress!
Footnotes
[1] Velligan, D. I., & Rao, S. (2023). The Epidemiology and Global Burden of Schizophrenia. The Journal of Clinical Psychiatry, 84(1). https://doi.org/10.4088/jcp.ms21078com5
[2] Singh K. K. (2013). An emerging role for Wnt and GSK3 signaling pathways in schizophrenia. Clinical genetics, 83(6), 511–517. https://doi.org/10.1111/cge.12111
[3] Komiya, Y., & Habas, R. (2008). Wnt signal transduction pathways. Organogenesis, 4(2), 68–75. https://doi.org/10.4161/org.4.2.5851
[4] Takahashi, H., Hashimoto, R., Iwase, M., Ishii, R., Kamio, Y., & Takeda, M. (2011). Prepulse inhibition of startle response: recent advances in human studies of psychiatric disease. Clinical psychopharmacology and neuroscience : the official scientific journal of the Korean College of Neuropsychopharmacology, 9(3), 102–110. https://doi.org/10.9758/cpn.2011.9.3.102
[5] Mulherkar, S., Uddin, M. D., Couvillon, A. D., Sillitoe, R. V., & Tolias, K. F. (2014). The small GTPases RhoA and Rac1 regulate cerebellar development by controlling cell morphogenesis, migration and foliation. Developmental biology, 394(1), 39–53. https://doi.org/10.1016/j.ydbio.2014.08.004
[6] Fortress, A. M., & Frick, K. M. (2015). Hippocampal Wnt Signaling. The Neuroscientist, 22(3), 278–294. https://doi.org/10.1177/1073858415574728
[7] Lisman, J., Yasuda, R., & Raghavachari, S. (2012). Mechanisms of CaMKII action in long-term potentiation. Nature reviews. Neuroscience, 13(3), 169–182. https://doi.org/10.1038/nrn3192
[8] FeaturedPsychology·October 16, & 2015. (2015, October 16). Inflammation in the Brain Linked to Schizophrenia Risk. Neuroscience News. https://neurosciencenews.com/neuroinflammation-schizophrenia-risk-2905/