The term “drugs of abuse” gets thrown around when trying to specify drugs that are detrimental in nature. This is opposed to the “drugs of use” that would include over-the-counter and prescription medication. It is far too often that these abusive drugs lead one into a path of addiction, and it is far too often that this path of addiction leads to death. So I must ask, why are “drugs of abuse” drugs of addiction?
Our remarkable human bodies have evolved in such fascinating ways. We, as a species, have honed in on the most practical ways to learn about the world, and remember what we learn, so that we might apply it in, ultimately, reproducing and raising our offspring. A result of this evolution is our ability to remember what gave us a “good” experience.
When I say “good”, I mean in the sense that the experience will increase one’s likelihood of creating and rearing successful offspring. This “good” experience can manifest itself in multiple ways: the awareness that comes along with a cup of morning coffee, the relaxation of a massage, the satisfaction in eating a pastry, or the intrinsic pleasure of sexual activity. It is such that all of these activities increase the odds of making successful children (some more directly than others).

Thanks to evolution, our brains devised incredible ways to “lock on to” these events, remembering exactly how they felt, and the specific steps taken to allow them to occur. Because of this, we get remarkably good at reproducing these feelings of pleasure, and most of the time, it is a very good thing.
The brain must transmit its signals somehow, and since the basic building blocks of life are chemicals, it chooses to transmit the signals as chemicals. Neurotransmitters, as they are called, are the chemicals released within the synapses of the brain, and they give us the ability to be humans. During the aforementioned “periods of pleasure”, many of these neurotransmitters are released, but the most important (and plentiful) of these is dopamine.

Dopamine is used colloquially as the “feel-good” hormone, and indeed it produces pleasurable effects. It is important in other aspects, however. As was mentioned, our brains are very good at remembering exactly how pleasurable events were created. It is dopamine that locks down these memories.
This is where drugs of abuse play such an important role. Nearly every abusive drug increases dopaminergic receptor agonism (“extra dopamine release”). This is either done in a direct (mescaline and amphetamine) or indirect (most other drugs of abuse) manner. Regardless of the process, the brain is receiving signals of pleasure from an unnatural source. Note, I use the term “unnatural” to mean that the brain is not triggering its own dopamine release, but rather substances from outside the body are.
Since these drugs can be administered in any dosage, it is quite common for internal dopamine levels to increase past the point of any natural setting. This incredibly high concentration of dopamine creates the “high” felt by abusers, as well as the second effect of dopamine: the formation of intense memories associated with obtaining the feeling of pleasure.
It is well-hypothesized that these “memory-forming” effects of dopamine play just as important of a role in addiction as the pleasurable effects do. While the pleasurable effects are likely more important in the development of withdrawal symptoms, the intense memories give addicts the urge that cannot be erased from their minds. When we look at the two together, we can see why the road to addiction is far too short.

Since extra dopamine is flooding the synapses, the brain thinks something is wrong, and down-regulates its dopamine receptors. This is, again, a result of evolution as it is meant to “reset” the brain to a normal state. But once the drug wears off, and dopamine levels return to normal, the brain cannot activate as many receptors as it normally would have. The result of this is initially depression, but follows with all sorts of horrendous symptoms which the reader may look up for themselves. This process is characterized as withdrawal.
The sad case of the matter is that eventually addicts do not seek the drug to experience a high, but rather to avoid a low.
The memory-effects of dopamine also come into play here, for just as the drug wears off, an abuser will remember the “good ol’ times” vividly. It will cease to matter whether the process of getting the drug is detrimental (expensive or dangerous); the addict will seek out a repeat experience nonetheless. And of course, each time the process “works”, the abuser will have strengthened the memory further. It is through this process that the “cravings” of addiction are created.
And so, terrible withdrawal symptoms, coupled with cravings, arises from repeated abuse of drugs. The well-known saying that drugs “hijack the body’s reward mechanism” are completely true. There is little escape from the self-administration of “dopamine” because of its grip on creating habits, and when administered in high concentrations, our bodies cannot prepare. What was meant to reinforce habits capable of creating life, in the end creates habits capable of destroying it.
ALS and the Contributions of Oxidative Stress
In the life disrupting disease known as amyotrophic lateral sclerosis (ALS), degeneration of upper and lower motor neurons eventually causes disability through the inability to control locomotion, speaking, eating and breathing. The most unfortunate parts of this disease are that the cause of ALS is not completely understood, treatments are limited with no cure, and ALS tends to progressively worsen over time even with treatment and therapy.

According to the ALS association, approximately 6,000 people in the U.S. are newly diagnosed with ALS each year. The average length of survival after diagnosis is three years, but there is much variability in length of survival as 5% of those diagnosed with ALS will live 20 years or more, like the famous physicist, Steven Hawking. Since ALS is such a debilitating disease, understanding of mechanisms of ALS and coming up with novel treatments will be essential to making a difference in the lives of those diagnosed with ALS.

This week we focused our learning on the paper: Oxidative stress and mitochondrial damage in the pathogenesis of ALS. This review identified 4 important possible contributors to ALS:
- Oxidative stress: occurs when a cell is unable to detoxify reactive intermediates, resulting in cellular damage. ALS tissues were found to have an accumulation of oxidative damage to proteins, lipids and DNA. This could be caused by a mutation in the SOD1 gene (superoxide dismutase), which normally acts as an antioxidant enzyme.
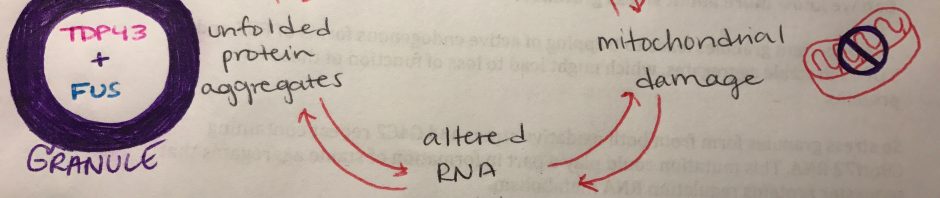
- Mitochondrial damage: oxidative stress can cause mitochondrial damage, creating energy deficits, calcium mishandling, and altering of RNA metabolism. Mitochondrial damage is especially important in motor neurons, which are known to have a large number of mitochondria.
- Altered RNA metabolism: Oxidative stress also causes the aggregation of RNA binding proteins (like TDP43 and FUS) into stress granules, which disrupt RNA metabolism. When RNA metabolism is disrupted, the overall health of the cell is compromised through the loss of functional proteins. Another important fact is that stress granule accumulation is common in patients with ALS.
- Unfolded protein aggregates: improper protein folding is a common to ALS pathogenesis. Stress granules may be trapping chaperones, proteins that assist in proper protein folding, having a widespread effect on cell physiology. In this situation, the protein degradation pathway is imbalanced, leading to the accumulation of misfolded proteins in ALS tissues. Improper folding of mitochondrial proteins will also affect mitochondria function!

These four cellular malfunctions found in ALS could all be impacting each other, resulting in a vicious cycle of damage to the important cellular organelles and processes that maintain a functioning motor neuron. While more research needs to be done in order to understand the complex pathways that contribute to ALS, the seeming overlap of oxidative stress and mitochondrial damage in ALS could be a potential target for future therapies to reduce the damage caused to motor neurons during the progression of ALS.
The Mystery of ALS: Motor Neurons
Amyotrophic lateral sclerosis (ALS) is a disease surrounded by a lot of mystery and frustration as the mechanisms of the disease are not fully understood, and there is no current way to cure, or even truly successfully treat the disease. ALS is a fatal disease characterized by degeneration of upper and lower motor neurons. Most patients with ALS end up dying of respiratory failure as their diaphragm is no longer able to function to help them breathe. Once patients are diagnosed, they are usually given a life expectancy of around two years. There is some hope though, as there have been certain people, like Stephen Hawking, who live for decades with the disease, we just have to figure out what to treat.
There are a couple main target points in the body that researchers have found to be associated with ALS. One is mutation of the SOD1 gene, which normally functions to protect the body from metabolic waste. ALS has been found to be associated with high oxidative stress in the body as well. This may be partly due to dysfunction of the SOD1 gene. It also appears to affect two RNA binding proteins, FUS and TDP43, not allowing them to properly function in RNA metabolism, affecting the functionality of other proteins in the body. The decreased function of these proteins may also lower the body’s ability to protect it from oxidative stress. Oxidative stress is also hurting mitochondrial function by affecting its proteins and metabolism. Thus, these two possible mechanisms seem to cross-over with each other.

One big question about the disease is why it only seems to cause degeneration of the motor neurons specifically?
I have come up with 5 possible reasons from looking at some research done by the ALS association:
1. Axon structure and proteins – motor neuron axons are around a meter long. This is a long way to transport messages, it would have high metabolic demands, and there is a lot of room for proteins associated with it to be affected making it harder to protect.
2. Too much cell apoptosis – even cell death that is programmed by the body can be harmful if it is happening too much. Possible halting some of the apoptotic events is being studied by scientists to possibly help the neurodegeneration.
3. Mitochondria – the motor neuron is huge and requires a lot of energy to stay functional. The mitochondria provides energy to its neuron, and as the mitochondria becomes dysregulated or dysfunctional, the motor neuron will lose its ability to function.
4. Glutamate – this is the main excitatory neurotransmitter of the body. Too much excitatory activity of a cell can lead to cell death, and in ALS there may be too much glutamate around and acting on the motor neurons.
5. Inflammation – this immune system process often happens as a result of cell death. If there is too much apoptosis going on or motor neurons or their surrounding cells dying from excitotoxicity, inflammation may be upregulated further damaging and killing more motor neurons.
Obviously there is no solid answer right now of the ALS disease mechanism. Maybe solving the “motor neuron mystery” will be key in treating ALS. It will be important to keep people aware of this disease, and keep research moving forward.
Quick Overview of ALS
Amyotropic Lateral Sclerosis (Lou Gehrig’s Disease) just might be one of the most terrifying conditions out there. For starters, at this point in research, 95% of cases are defined as sporadic. That essentially means that there is no known cause or biomarker that can determine one’s susceptibility of having ALS in the future.
Oxidative Stress and ALS
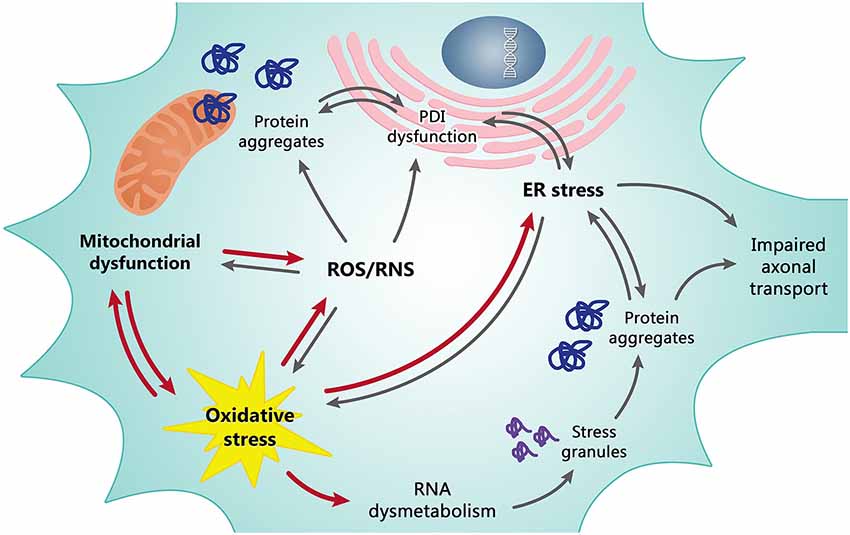
Motor neurons become bombarded with oxidative stress from substances such as radical oxygen species which leads to multiple different responses from the cell. SOD1 which is an enzyme that captures these radical oxygen species and converts them into a manageable molecule that can be destroyed within the cell. In some cases of ALS, SOD1 enzymes are not properly formed, allowing the radical oxygen species roam within the cell causing oxidative damage.
Stress Granules

When certain cells are exposed to oxidative stress they begin to form complexes of proteins and untranslated mRNA to become what is called a stress granule. These granules can perform several different tasks such as signaling, the decay of RNA, and apoptosis (cell death). Stress granules may seem like they could be a problem because they can lead to cell death, but they are a designed protective measure by the cell that helps protect the cell while it is experiencing oxidative stressors. The problem comes from the overabundance of stress granules due to prolonged oxidative stress. This leads to a build-up of granules blocking other cellular processes leading to further dysfunction within the cell.
Big Picture?
So, what does all this oxidative damage do? Over time these motor neurons are killed by the prolonged damage leading to a lack of signal from the brain to various muscles. The slow deterioration of motor neurons leads to muscle weakness and slowly ends up in paralysis of muscles. The scary part is that there isn’t necessarily a defined path that ALS follows in which muscle usage deteriorates. Furthermore, many may not realize that they have ALS symptoms because the age of onset doesn’t come until people are in their 50s (average) so they may view these symptoms as a normal process of aging.
Prevention and Treatment
This is the more disheartening aspect of ALS because there is no surefire treatment that has extended the patient’s life significantly. There is some recent research into using antioxidants to help slow down the progression of ALS but they are too recent to find out the results of the study. This seems to pose the question, “What do I do to prevent myself from getting ALS?” Some may say that there is no way to prevent ALS from happening, but I would venture to assert that antioxidants wouldn’t hurt. Clearly, we are missing a piece that might help us figure out the cure but that might be further down the road so at this point the best bet is to live life to the fullest until the inevitable comes.
Does ALS Stand for All Is Lost Suddenly?
Does ALS Stand for All is Lost Suddenly?
Amyotrophic lateral sclerosis, better known as ALS or Lou Gehrig’s disease, is a central nervous system disease where motor neurons are targeted. The motor neurons send signals to your muscles all over your body, even ones used to speak, swallow, and breathe. According to the Mayo Clinic website, ALS symptoms begin with muscle weakness in your hands, feet, and limbs, and continues to spread over time. As the disease progresses, the more vital processes such as breathing and speaking make living with the disease very difficult. Individuals that are diagnosed with ALS typically are told they have only a couple years left. However, this does not mean that it will always be the case. Genius Stephen Hawking has lived 50 years with ALS. So what causes this awful disease? What hope is there for those who are unfortunately diagnosed?
As of today, there is no certain answer as to what is the official cause of ALS. However, researchers have determined a few processes that are present in ALS patients. These processes are as follows:
Oxidative Stress and Mitochondrial Damage
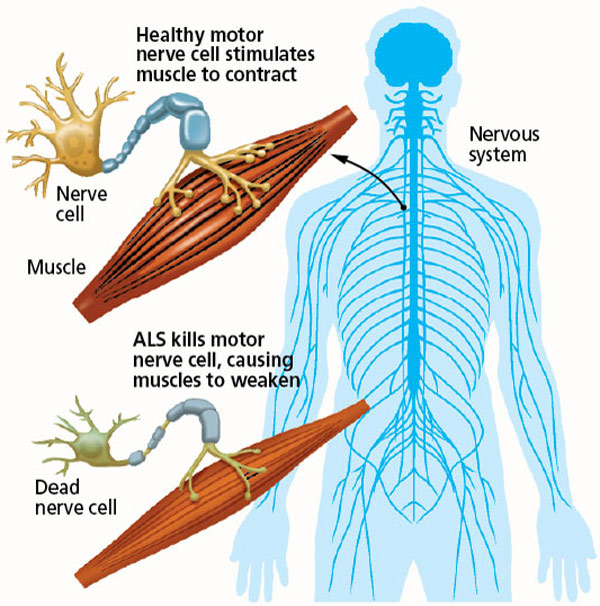
Oxidative stress comes from a high amount of free radicals and not enough response to break down these free radicals. The body naturally has antioxidants and enzymes to regulate these free radicals, however, in ALS patients, the enzyme SOD1 typically used to relieve oxidative stress is mutated and cannot function properly. This oxidative stress causes the mitochondria to become damaged and RNA to be incorrectly spliced/metabolized. Below is an image of a motor neuron. The mitochondrion can be seen labeled toward the bottom (the little tan bean shaped organelles).

RNA Dysmetabolism
There are two RNA binding proteins called FUS and TDP43. Both of these proteins are supposed to be in the nucleus of a cell and interact with RNA to help in making more proteins properly. However, in ALS patients, FUS and TDP43 are removed from the nucleus and accumulate in the cytoplasm. Since they are no longer in their proper location, the proteins that they would have helped produce would no longer be made correctly, causing mayhem in the mitochondria. Since oxidative stress is highly localized in the mitochondria, the relocation of FUS and TDP43 can be linked to the cause of oxidative stress and mitochondrial damage.
Protein Folding and RNA Binding Proteins
FUS and TDP43 also have an effect on protein folding. There are certain chaperone proteins that allow other proteins to be imported into the mitochondria and allow the proteins to fold correctly. However, FUS and TDP43 can actually trap the necessary chaperones which does not allow the proteins to get where they need to be and are not folded correctly. If proteins are not folded correctly, they are unable to do their jobs and must be broken down by the cell. If there are too many misfolded proteins in the mitochondria, oxidative stress builds and the mitochondria is damaged, and RNA is not metabolized correctly.
As we can see, it seems that each of these causes discussed stems from and causes the others. Unfortunately because of this, no one knows where ALS truly originates, and it is almost impossible to know what exactly to target to treat this disease. One drug does exist that only extends life for 2-3 months, but this drug also costs $14,000.
It is never easy to hear a loved one has been diagnosed with an illness that has no known cause or cure. Coping can be difficult and your life is forever changed. I am confident, however, that our modern medicine and advancing research will, one day, find a way to treat or even cure this most unfortunate disease. For now, all we can do is hope. All is not lost suddenly.
What Science Doesn’t Know – ALS
Amyotrophic Lateral Sclerosis (ALS) – also commonly known as Lou Gehrig’s Disease – is a degenerative disease that can effect motor neurons in the central or peripheral nervous system. This leads to a steady loss in motor skills -including coordination and movement of skeletal muscles. Sometimes dementia can form even if it isn’t a common symptom. Eventually, death will occur when the degeneration of motor neurons begins to affect basic functions such as breathing, swallowing, and heart rate (1). Unfortunately ALS is an incurable disease with no known cause or treatment at this point in time.Furthermore, there are currently no known indicators for ALS onset. In fact, diagnosis of ALS often is the result of ticking off what other diseases it could be.
Which brings up the point: why is so little known about ALS?
This question is especially frustrating considering the harsh reality of the disease. And like with most diseases the answer is complex and multifaceted. The matter of the underlying causes of ALS isn’t necessarily just about what can lead to ALS – but what doesn’t contribute to ALS. Numerous studies have linked glutaminergic excitoxicity, genetics, immune dysfunction, misfolding of proteins, environmental toxins, and even military service to the development of ALS (2). The disease is also known to occur more frequently in men. So it’s quite difficult to pinpoint any given cause to the development of ALS let alone find an effective treatment. However, that doesn’t mean that a cure or treatment for ALS can’t be achieved.
If you wish to donate to ALS research you can give any amount to the ALS Association (no Ice Bucket Challenge required).
Sources:
- http://www.webmd.com/brain/tc/amyotrophic-lateral-sclerosis-als-topic-overview#1
- http://www.mayoclinic.org/diseases-conditions/amyotrophic-lateral-sclerosis/symptoms-causes/dxc-20247211
So You Did the Ice Bucket Challenge, Now What?
Along with 2.4 million other people, I shared a video of myself doing the ALS ice bucket challenge on Facebook in the summer of 2014. I excitedly watched ice bucket challenge videos go viral and waited for my turn to get tagged in one so I could participate and be a part of the “in crowd.” I willingly participated when I was challenged and tagged a handful of friends and family members to pass it on when my turn came and that was the last I thought of it

I have a feeling my ice bucket challenge narrative might sound familiar to some. Prior to doing the challenge, with neuroscience among my majors, I was familiar with amyotrophic lateral sclerosis (ALS) and knew it was a neurodegenerative disease but I didn’t really think much about it. I knew it struck primarily men, and thought I was safe, since I did not even know females could get ALS.
The fact of the matter is, ALS is perhaps the most devastating disease I can think of. Symptoms come quickly, one day you’re a healthy 28-year old, the next day you feel a bit weak and tired, and within just a couple of years, your motor neurons in your spinal cord have completely died, leaving you paralyzed, but still completely aware of your surroundings. With no curative treatment, average life expectancy for someone diagnosed with ALS is 2-5 years. The single approved treatment only prolongs life by a couple of months and costs thousands of dollars.

ALS is characterized by the degeneration and death of motor neurons in the spinal cord. While the reason for the cell death is unknown, it is thought that oxidative stress in our cells brought on by free radical molecules cause the mitochondria (responsible for making cellular energy) to become defective and send the cell down the death pathway (apoptosis). In addition, RNA (the intermediate between DNA and protein products) is found to be deregulated in ALS, which in turn causes more cell stress, mitochondrial damage, and leads to apoptosis.

The truly terrifying thing about ALS is that you can do absolutely nothing to avoid getting it. I’d like to think I’m a fairly healthy person, I don’t smoke, I exercise, and I eat my vegetables. While this might be great to avoid cancer, if I’m going to get ALS, I’ll get it regardless of anything I do or do not do. There is also no way to predict who will get ALS. There is no magic “ALS gene” that you can test for if a family member has ALS. While familial ALS is a type of ALS, it only makes up 5% of cases of ALS, leaving sporadic ALS to make up 95% of cases, leaving it totally up to chance.
The good news is that there is good news. In 2014, the ice bucket challenge raised $115 million for the ALS foundation. Of that, $3 million went to fundraising efforts, $10 million went to public and professional education, $23 million went to patient and community services, and $77 million went to research for ALS. This astounding amount of money dedicated to research assisted in identifying a new gene thought to be implicated in about 3% of ALS cases, NEK1. While 3% doesn’t seem to be that much, it a larger percentage of cases than any of the 20 other genes implicated in ALS. While not much is known about NEK1, since it was only recently identified as being implicated in ALS, it is thought to be involved in cell cycle regulation, which can tell a cell when or when not to divide or die.

Until scientists find a cause, conclusive test for ALS, and a cure, the most we can do is support patients with ALS and their families by continuing to raise money and awareness for ALS. Talk about ALS, participate in future ice bucket challenges, and maybe donate some money to the ALS Foundation. Visit the ALS Foundation website for ways to get involved, including the ALS Foundation walk hosted in Fargo/Moorhead on August 19, 2017.
ALS Foundation website: http://www.alsa.org
Diagnosing and Dealing With ALS
A diagnosis of ALS is often accompanied by feelings of fear, frustration, sadness, and anger. Amyotropic lateral sclerosis (ALS) affects about 6,000 Americans every year and usually appears in midlife. In order to better cope with a diagnosis or help a loved one going through it, education is important. Understanding what we do know about the disease can be beneficial. The image below outlines some of the potential contributing factors to ALS.
ALS can be difficult to clinically diagnose because there is no test for it. It is diagnosed by ruling out other options such as: thyroidtoxicosis, cervical spondylosis, diabetes affecting brachial plexus, carpal tunnel syndrome or lesion on the elbow, neuropathy, chronic inflammation, myasthenia gravis, sclerosis, spastic paraplegia. Only about 10-15% of people have a false positive diagnosis.
After a diagnosis you visit the doctor every 3-6 months. At an ALS Association Certified Center of Excellence, your team typically consists of the neurologist, RN, speech pathologist, respiratory therapist, physical therapist, occupational therapist, dietician, rehab tech, and a social worker. You can participate in clinical trials for new drug treatments if you fit the criteria.
The current understanding of the pathology of the disease is limited and there are many factors affecting the onset of ALS. We know that oxidative stress causes changes in the cell that contribute to the death of motor neurons. As you can see in the drawing, in the presence of oxidative stress, stress granules form and remove TDP43 and FUS from the nucleus (information center) of the cell. TDP43 and FUS are critical to cell function, and when they are removed from the nucleus and clump up in the cell, it causes damaging effects seen in ALS patients.
Treatment of ALS is very difficult to find because of the complicated nature of the disease. Researchers have not been able to find the key target that would stop or reverse the effects of the disease. The current drug on the market Riluzole only prolongs life by 2-3 months and is an antiglutamate drug acts by reducing levels of excess glutamate found in the nervous system. Current clinical trials are in progress for trying to use stem cells to promote neuron survival and growth. Because 90-95% of cases are sporadic it is difficult to pin down a genetic sequence that is inherited to explain the cause of the disease. It also tends to be found in Caucasians and is more common in males than in females.
While this disease is scary, it needs to be talked about to raise awareness and help work toward finding a cure. In order to better cope, it is helpful to join a support group and learn as much as you can about the disease. It is recommended to maintain a good lifestyle, healthy eating, exercise and time with loved ones.
For more information see: http://www.alsa.org/
ALS, the Battle for the Nervous System

Amyotrophic lateral sclerosis (ALS), or Lou Gehrig’s disease, is a neurodegenerative disorder of motor neurons. Motor neurons are a type of nerve cell that stimulates muscles so they can move. Motor neurons can originate from the brain or spinal cord. You’ve likely heard about this disease if you are familiar with Stephen Hawking or have read the short novel Tuesday’s with Morrie. Although Stephen Hawking has lived to be 75 years old, the average person with ALS dies only 3 years after being diagnosed. Symptoms of ALS include the following:
- Difficulty performing daily activities like walking
- Impaired speech
- Weakness or clumsiness of hands and/or feet
- Muscle cramps

Our neurochemistry class at Concordia College focused on reading literature on the underlying mechanisms behind the cause of ALS. Scientist are now finding that oxidative stress on the motor neurons could be the source of the mysterious die off of cells. They are hypothesizing that a sub set of people are lacking a few ‘house keeping’ proteins that normally allow motor neurons to deal with an increased work load. Without these essential proteins, the neurons eventually die because of the toxic build up of metabolic by-products.
One of the most frustrating parts of neurodegenerative diseases is that there are no cures for any of them at this time. For the people not involved in the research as well as those who have been diagnosed with ALS or any of the other myriads of neurodegenerative diseases, it can be easy to rationalize anger towards the scientific community for not solving this problem yet. You’re telling me we have the capability of performing a head transplant, but we don’t know how to cure a single nervous system disease??
I had this same thought a few years ago before I attended the national convention for the Society of Neuroscience. Then I saw the work being done behind the scenes. Hundreds upon hundreds of presentations on every sort of nervous system disease (an entire section dedicated to ALS studies) you could possibly name. Undergraduates like myself, graduate students presenting their dissertations, as well as professors that have dedicated their lives to understanding diseases like ALS showed the information they had gathered.
Despite the largest convention center in North America being filled to the brim with the newest information on nervous system diseases, we still don’t know what causes ALS, nor do we understand how it manifests in ~5,500 new people every year. With the vast amount of knowledge we have, the question is raised of whether or not we will ever find a cure? Could a cocktail of synthetic drugs leave us in peace for our remaining years? Or are we now actually trying to battle mortality? The disease that used to plague us consisted of bacteria and viruses that attempted to take over our bodies, but now we are battling the wear and tear of life. Maybe we will find a cure for ALS. Then again, maybe we’re not meant to. Regardless of future scientific advances, we should do our best to live every day to the fullest. Life is precious because it doesn’t last forever.

Where Do We Draw the Line on Research?
Just three weeks ago, I purchased A Brief History of Time, by Stephen Hawking, and less than two weeks ago, I finished the book. From what I hear, Stephen Hawking’s persona is equally linked to ALS (amyotrophic lateral sclerosis) as it is to black holes, quantum mechanics, and general relativity. The “pop culture” idea of the man is as the wheelchair-bound genius. Thus, I found it remarkably coincidental that the following Monday I was required to read a recent journal review of ALS for my neurochemistry course.

The review highlighted recent research regarding RNA dysregulation and oxidative stress as main culprits for the debilitating disease. It insisted that the two were inexplicably linked through the processes of two nuclear (i.e. pertaining to the nucleus of the cell) proteins, FUS and TDP43. The overarching theory was that oxidative stress would cause these proteins to leave the nucleus, and find their way to other parts of the cell where they would cause problems with RNA (think of it as the link between DNA and proteins). Basically, oxidative stress was destroying the cell’s ability to properly make proteins. This would in turn lead to more oxidative stress which would just start the cycle over again, eventually the cell (a motor neuron, the body’s way of telling muscles to move) would die, and thus we have ALS.
This is truly a remarkable theory, and certainly there is evidence to support it, but there is a competing theory: excitotoxicity of motor neurons. Excitotoxicity is a big word. It means that the cell is getting too much stimulation, and thus eventually dies. (Too much of anything is bad it may seem.) Indeed, the best pharmaceutical drug prescribed to ALS, riluzole, is a glutamatergic inhibitor (prevents excitotoxicity). However, even this drug is thought to extend the average life expectancy of a patient, which is only 2-3 years usually, by only a few extra months.

If the drug works at all, it must be targeting something that is causing the disease, so we can readily assume that excitotoxicity is a culprit. But the drug certainly does not cure ALS, so it cannot be giving us the full story. Perhaps the review I mentioned gives the other half of the story, though that is highly doubtful. In the end, we must agree that there is no single answer to solving this problem.
I then look back at Hawking, and his book, A Brief History of Time, and again notice a coincidence. Hawking argues that our knowledge of the universe is likewise incomplete, as there may indeed be no singular answer to the problem. He argues for more collaboration, and ultimately more research. If this is to be the case, however, I wonder if perhaps our research into the workings of the universe would be misplaced.

Let us imagine, for a second, what kind of world we would live in if instead of wasting hours on end mathematically modeling the trajectories of black holes spiraling into one another, we put our efforts into saving lives medically. We spend enormous amounts of time on understanding the maddeningly large, and the unthinkably small, that we forget about the in-between where lives may be saved. How is it that we can say how much slower time may progress for a relativistic traveler, but we cannot discover the causes of ALS? Could Hawking himself have, if not cured, perhaps contributed to his own disease’s salvation? Educated human beings place so much effort into things that will likely never save lives. And I constantly wonder if my own pursuit of mathematics will do “harm” to the world in the sense that I am not doing as much “good” as I could in chemistry.
Without a doubt, we (as a species) could have saved millions of lives from countless diseases if we had put our efforts into medical technology, instead of other pursuits. And yet we continue to do otherwise. Why? Indeed, there can only be one answer. Human lives cannot be quantified as “better” by the amount of time they exist. What I mean to say is, saving a life is not saving a life if one has instead thrown away one’s own life in the process. We are only here for a fleeting moment, and as such it is important for each of us to make the most of the fleeting moment. That may indeed consist of saving lives medically, but it may also consist of “saving lives” in other ways. Perhaps an article written by a journalist prompts a young boy to enter politics, wherein he pushes to allocate more money to NASA, who in turn create a nano-scale polymer to shed heat on rockets reentering the atmosphere, but indeed that nano-scale science is published and gives medical researchers the ability to create cancer-targeting drugs, wherein countless lives are saved. The journalist, perhaps dead for twenty years, has just saved lives, by doing what she loved best. A scenario like this, can be represented by the butterfly effect, which states that the smallest change in initial conditions can have unforeseeable consequences for the future.
In the end, then, we can at least agree that our actions, however we decide to make them, will certainly change, and possibly even save, lives. Stephen Hawking may likely still be bringing about his disease’s salvation, just in an obscure way. But at the same time, he is following his own path and immensely impacting the world for the better. It is not only our right, but our duty to pursue our own passions, even at the extent of others’ deaths.