Feature Image from Pexels
Reflecting on my Semester in Neurochemistry:
This semester in neurochemistry has been one of the most challenging yet rewarding experiences of my time at Concordia. Entering the course, I expected to learn about brain signaling and function, but I did not anticipate how deeply the material would shape the way I think about my own life. Throughout the semester, my learning evolved from strictly memorizing the molecules in each pathway to interpreting scientific research and learning how to connect these complex biological mechanisms to real-world issues. Rather than simply understanding concepts at a surface level, I learned how to critically evaluate research articles, identify key findings, and discuss future implications as both a scientist and citizen.
In addition to analytical skills, I also developed stronger communication abilities. Writing blog posts throughout the semester forced me to translate dense scientific information into clear language the general public would understand and engage with. This process helped me realize that truly understanding a concept means being able to explain it to others. It also made me more aware of the importance of science communication among the general public, especially when discussing topics like cancer, mental health, and the effects of substances on the brain.
Neurochemistry’s Role in Becoming Responsibly Engaged in the World:
Neurochemistry has fundamentally changed how I think about what it means to be responsibly engaged in the world. Understanding the biological basis for behaviors has made me more empathetic and more aware of the factors that influence people’s actions and health. For example, learning about the neurochemical mechanisms of addiction highlighted that it is not simply a matter of personal choice, but a complex condition influenced by brain chemistry, the environment, genetics, and cultural beliefs.
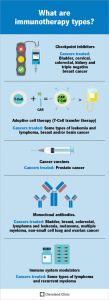
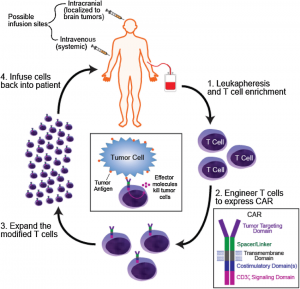
This perspective aligns closely with Concordia’s goal of encouraging responsible participation in the world. With a deeper understanding of these issues, I feel much more equipped to engage in informed discussions about public health and ethical policies. Neurochemistry has also emphasized the importance of scientific literacy in today’s society. Misinformation about health and science is rapidly increasing, so having the skills to critically evaluate such claims and evidence is critical. Topics like cancer treatment, pharmaceutical development, and the use of cannabinoids and psychostimulants raised important questions about accessibility, regulation, and long-term effects. These discussions reinforced the idea that being responsibly engaged in the world involves not only understanding scientific concepts but also considering their broader social and ethical implications.
My Development of Foundational Skills and Interdisciplinary Perspectives:
Another core goal of Concordia’s liberal learning framework is the development of foundational skills and transferable intellectual capacities, and this course contributed significantly to that. Overall, I strengthened my ability to analyze experimental data, interpret intricate scientific literature, and develop evidence-based arguments for discussion. Furthermore, these skills are not only limited to neurochemistry. Rather, they can be applied across all disciplines and will be valuable in any future academic or professional setting I find myself in.
Another important aspect of my intellectual development was gaining an appreciation for interdisciplinary perspectives. Neurochemistry is inherently connected to multiple fields, including psychology, biology, and pharmacology. For instance, when studying topics like obesity or mental health disorders, it became clear that understanding the brain requires more than just biochemical knowledge. It also involves considering behavioral patterns, environmental influences, and cultural factors.
What Liberal Learning Means to Me:
Another one of the five goals of liberal learning is to instill a love for learning, and I can confidently say that this course has contributed to that goal. Despite the challenges, I found myself genuinely interested in the material and was motivated each week to explore each topic deeper.
Through studying neurochemistry, I have also gained a deeper understanding of my own behavior and experiences, which has influenced how I approach my health and well-being. It has also encouraged me to think more critically about the ethical questions of scientific research and medical practices.
Ultimately, liberal learning means becoming a more well-rounded individual who is prepared to engage with the complexities of the world. It is not just about acquiring knowledge, but about developing the skills and perspectives needed to use that knowledge responsibly.
Relation to My Future Goals:
The skills and knowledge I gained this semester also support my goal of becoming a physician assistant. Neurochemistry strengthened my understanding of how biological processes influence health and disease, which is essential for patient care. As a future PA, I will need to evaluate medical conditions and contribute to treatment decisions, and this course helped build a strong foundation for such responsibilities.
I also improved my ability to interpret and communicate scientific research. Analyzing articles and writing blog posts taught me how to break down complex information clearly, which is an important skill for collaborating with my fellow healthcare team and explaining conditions to patients.
Most importantly, this course emphasized the importance of an interdisciplinary approach. Understanding the connections between biology, psychology, and social factors will help me provide more holistic and empathetic care. Overall, this experience strengthened my critical thinking skills and reinforced my motivation to pursue a career as a physician assistant.
One Last Conclusion:
Reflecting on this semester, it is clear that this neurochemistry course has been a significant part of my liberal learning experience at Concordia, as it has changed the way I approach learning in and of itself. Overall, I am leaving this course not only with a deeper understanding of neurochemistry, but also with a stronger appreciation for lifelong learning.
As I transition into my final year of undergrad, I will continue to use the skills I have gained. Whether through my future career, continued education, or everyday interactions, I hope to apply what I have learned to contribute positively to the world around me. In that sense, this course has not only been an academic experience, but also an important step in becoming responsibly engaged in the world and truly embodying the goals of liberal learning.