The brain has its own cannabis system, and long before humans cultivated marijuana, our neurons were already communicating using cannabis-like molecules called endocannabinoids.
These molecules help regulate mood, memory, appetite, pain, and synaptic plasticity. They act as a kind of neural “volume control”, fine-tuning communication between neurons. This internal system, called the endocannabinoid system (ECS), is essential for maintaining balance in the brain.[1] Under normal conditions, it works beautifully. But what happens when that carefully balanced system is repeatedly overstimulated by external THC? This question sits at the center of current cannabinoid research.
CB1 Receptors and Neural Signaling
In their review, Kendal & Yudowski (2017) describe how CB1 receptors – the primary receptors activated by THC – are among the most abundant G-protein-coupled receptors (GPCRs) in the brain.[1] These receptors are densely expressed in the hippocampus, prefrontal cortex, basal ganglia, and cerebellum – regions responsible for memory, executive function, reward, and movement.
Under normal conditions, endocannabinoids are produced on demand. They briefly suppress neurotransmitter release to fine-tune synaptic signaling. THC, however, is not produced on demand. It persistently activates CB1 receptors.
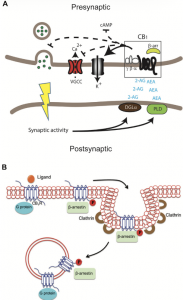
Figure 1 (left): Mechanism of retrograde endocannabinoid (eCB) signaling via CB1 receptors in a neural synapse [1]
Kendall & Yudowski explain that repeated THC exposure leads to CB1 receptor desensitization – meaning the receptors become less responsive over time.[1] This contributes to tolerance. The brain adapts by reducing signaling efficiency, shifting intracellular signaling pathways, and altering neural communication. In other words, the brain compensates.
How THC Changes Brain Function
In the short-term, THC exposure is associated with a plethora of cognitive changes, including impaired short-term memory, reduced attention, slower reaction times, altered motor coordination, as well as anxiety. These effects are largely explained by CB1 receptor activation in the hippocampus and prefrontal cortex. For most users, these impairments are temporary, but intoxication can increase risky behavior like impaired driving.[2]
On the other hand, chronic overstimulation of CB1 receptors with repeated or early exposure can produce tolerance, increase the risk of heart disease or stroke, alter reward circuitry, and disrupt executive function.[3]
Adolescent exposure may be particularly concerning. The ECS plays a role in synaptic pruning and cortical maturation during development. Introducing THC during this window may interfere with normal circuit refinement.
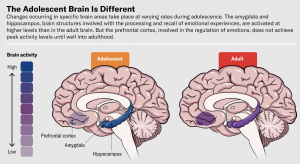
Figure 2: The progression of prefrontal cortical maturation and synaptic refinement during adolescence. [4]
Epidemiological studies have linked heavy adolescent cannabis use to increased risk of cognitive impairment and psychiatric vulnerability. [5]
What This Means for Us
While cannabis legalization is expanding, the perception of harm is decreasing, and THC potency has increased dramatically over the past few decades. We now know that the ECS system is not just another neurotransmitter pathway – it is a central regulator of synaptic balance. Repeated exogenous stimulation, however, may shift that system away from homeostasis. So understanding how THC interacts with CB1 receptors is not about morals; it is about informed decision-making. Science does not suggest that all cannabis use leads to irreversible harm. It does suggest that timing, frequency, dose, and developmental stage matter. The ECS system evolved to maintain balance, but the real question remains, “What happens when balance becomes overstimulation?”
References
[1]
D. A. Kendall and G. A. Yudowski, “Cannabinoid Receptors in the Central Nervous System: Their Signaling and Roles in Disease,” Front. Cell. Neurosci., vol. 10, Jan. 2017, doi: 10.3389/fncel.2016.00294.
[2]
“Marijuana,” Cleveland Clinic. Accessed: Feb. 24, 2026. [Online]. Available: https://my.clevelandclinic.org/health/articles/4392-marijuana-cannabis
[3]
N. D. Volkow, R. D. Baler, W. M. Compton, and S. R. B. Weiss, “Adverse Health Effects of Marijuana Use,” N Engl J Med, vol. 370, no. 23, pp. 2219–2227, Jun. 2014, doi: 10.1056/NEJMra1402309.
[4]
B. C. Meyer Heidi, “Brain Science Has Discovered New Drug-Free Approaches for the Anxious Adolescent,” Scientific American. Accessed: Feb. 24, 2026. [Online]. Available: https://www.scientificamerican.com/article/adolescent-anxiety-is-hard-to-treat-new-drug-free-approaches-may-help/
[5]
M. Arain et al., “Maturation of the adolescent brain,” Neuropsychiatr Dis Treat, vol. 9, pp. 449–461, 2013, doi: 10.2147/NDT.S39776.



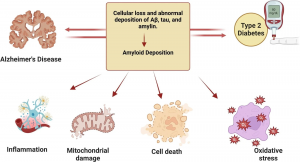
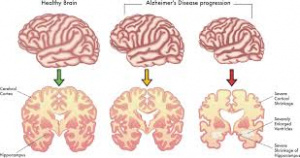

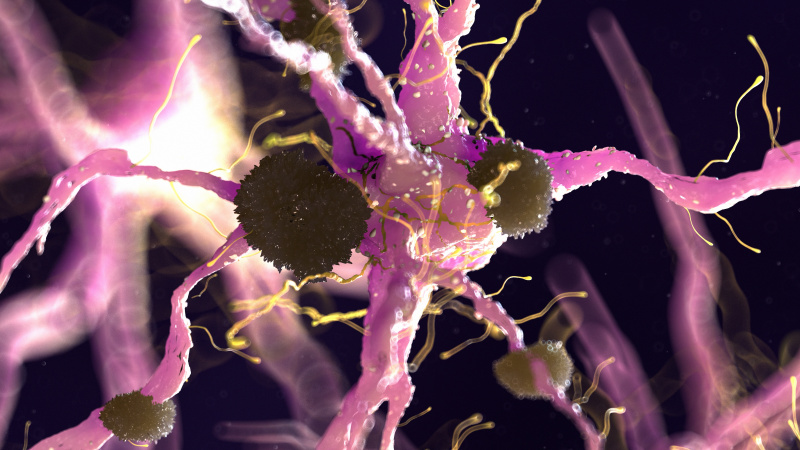
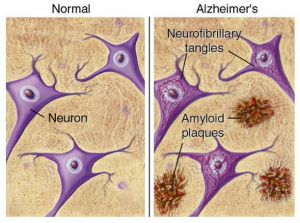
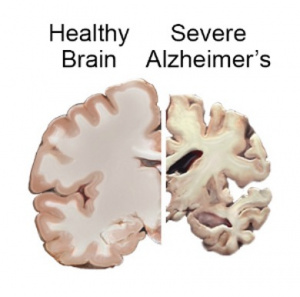
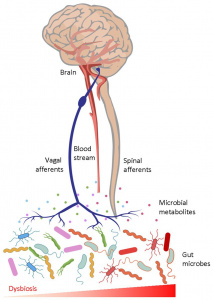
 Interestingly, 80% of Alzheimer’s patients also have type 2 diabetes. When we eat an excess amount of calories, our cells adjust the amount of receptors in order to avoid overactivation of insulin pathways caused by the high levels of insulin released. Highly processed foods are characterized by high levels of sugar and unhealthy fats which are extremely calorically potent and therefore they increase insulin resistance (3). By increasing insulin resistance, we make it more difficult for insulin to do its job, which consequently results in less activation of PI3K and therefore less deactivation of GSK-3B. Additionally, diets that are high in fat can result in chronic neuroinflammation, which also contributes to AB plaque accumulation and NFT formation (4).
Interestingly, 80% of Alzheimer’s patients also have type 2 diabetes. When we eat an excess amount of calories, our cells adjust the amount of receptors in order to avoid overactivation of insulin pathways caused by the high levels of insulin released. Highly processed foods are characterized by high levels of sugar and unhealthy fats which are extremely calorically potent and therefore they increase insulin resistance (3). By increasing insulin resistance, we make it more difficult for insulin to do its job, which consequently results in less activation of PI3K and therefore less deactivation of GSK-3B. Additionally, diets that are high in fat can result in chronic neuroinflammation, which also contributes to AB plaque accumulation and NFT formation (4).
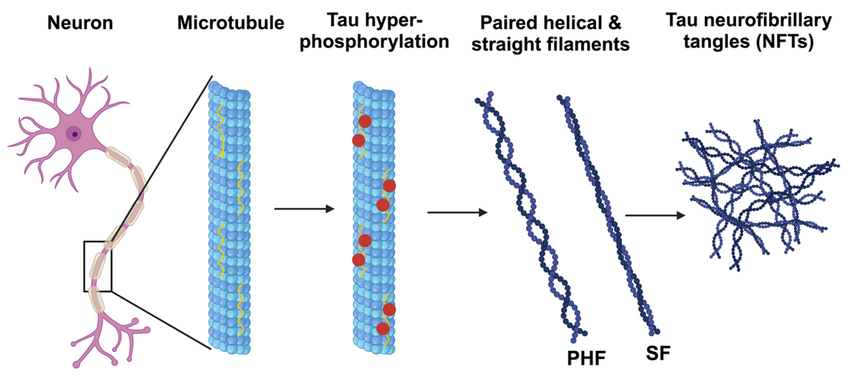

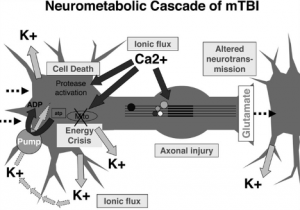 Figure 1: Neurometabolic cascade following concussion[2]
Figure 1: Neurometabolic cascade following concussion[2]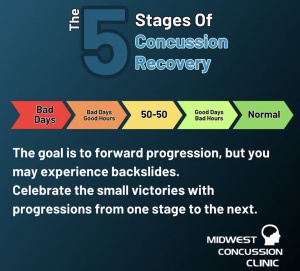 Figure 2(right): Illustration of the nonlinear nature of concussion recovery, showing that progress may fluctuate and reinforcing the importance of considering biological healing beyond symptom resolution.[3]
Figure 2(right): Illustration of the nonlinear nature of concussion recovery, showing that progress may fluctuate and reinforcing the importance of considering biological healing beyond symptom resolution.[3]