artstract by Olivia Pederson
What is a Fad Diet?
 We have all heard and seen that the south beach diet, the keto diet, the 3-day diet, the juice diet, and many more of these types of diets are recommended for fast weight loss, but this is 100% wrong. These diets are all called ‘fad diets’ which mean that they are popular for a short time, have no standard dietary recommendation, and made pseudoscientific claims for fast weight loss. These diets are not supported by clinical research and often make unsubstantiated statements about health and disease.
We have all heard and seen that the south beach diet, the keto diet, the 3-day diet, the juice diet, and many more of these types of diets are recommended for fast weight loss, but this is 100% wrong. These diets are all called ‘fad diets’ which mean that they are popular for a short time, have no standard dietary recommendation, and made pseudoscientific claims for fast weight loss. These diets are not supported by clinical research and often make unsubstantiated statements about health and disease.
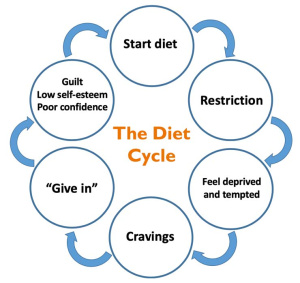 45 million Americans go on diets each year, and roughly 50% of them use fad diets. These diets usually only last a couple weeks to a couple months, but once the diet is over it is easy to fall back into the way people were eating before. This turns into a diet cycle that doesn’t end. This cycle can lead to poor health outcomes such as eating disorders, malnutrition, anemia, metabolic abnormalities, and impaired quality of life.
45 million Americans go on diets each year, and roughly 50% of them use fad diets. These diets usually only last a couple weeks to a couple months, but once the diet is over it is easy to fall back into the way people were eating before. This turns into a diet cycle that doesn’t end. This cycle can lead to poor health outcomes such as eating disorders, malnutrition, anemia, metabolic abnormalities, and impaired quality of life.
New Years Resolutions and Social Media
 The beginning of the new year brings up this idea of new years resolutions. January is a very common month to see these new diet trends make an appearance as well as weight loss products. In the United States alone, 80% of people fail their new years resolutions by February. This is done by not following healthy lifestyle changes over an extended period of time.
The beginning of the new year brings up this idea of new years resolutions. January is a very common month to see these new diet trends make an appearance as well as weight loss products. In the United States alone, 80% of people fail their new years resolutions by February. This is done by not following healthy lifestyle changes over an extended period of time.
Social media plays a large role in diet culture and fad diets. These social media platforms influence many peoples lifestyle behavior or social norms. These fad diets are usually promoted by celebrities or by people who are a part of a company to sell a product. Many of these diets and products promoted for weight loss are not evidence-based. Social media also portrays unrealistic body standards to people, especially young people. There are always people posing or flexing their body instead of relaxing and showing what they actually look like, which can really effect how people see their own bodies. It is important to take each social media post with a grain of salt because it is more than likely not accurate.
The Science Behind Obesity
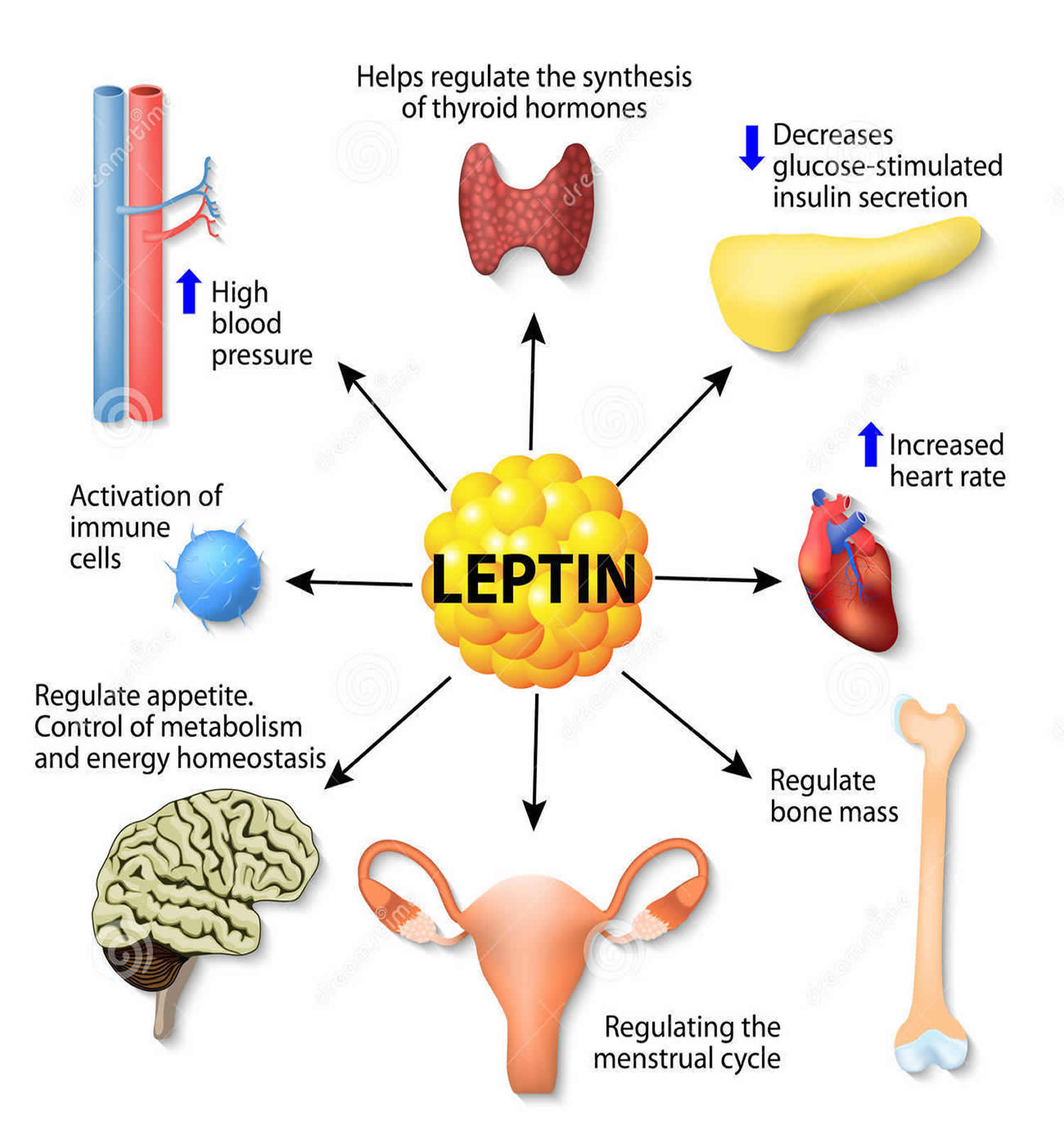
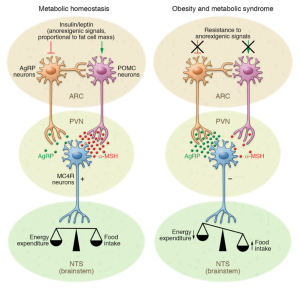 The hypothalamus in the brain controls a number of neuroendocrine functions that integrate metabolic feedback and control energy balance. Insulin plays an important role here because it is used to help turn food into energy. Leptin is another important character here because it is a hormone released from fat cells and regulates long-term energy balance. These two signals are essential in hypothalamic control of energy balance.
The hypothalamus in the brain controls a number of neuroendocrine functions that integrate metabolic feedback and control energy balance. Insulin plays an important role here because it is used to help turn food into energy. Leptin is another important character here because it is a hormone released from fat cells and regulates long-term energy balance. These two signals are essential in hypothalamic control of energy balance.
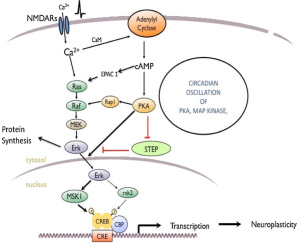
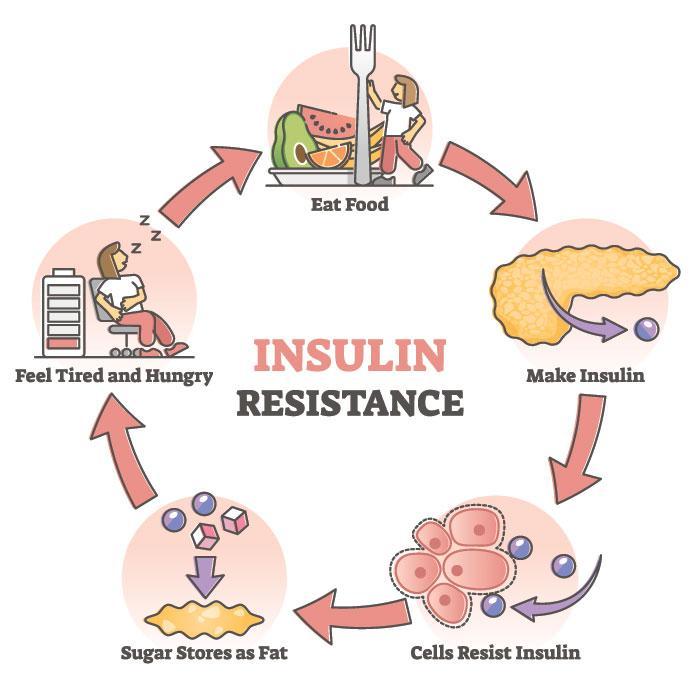
The image above shows this mechanism. The hypothalamus senses and integrates feedback from certain hormones that circulate in the body. Insulin and leptin act on neuronal subsets of the hypothalamus to control energy balance. Through certain excitation and inhibition of specific neurons, energy usage and food intake is balanced out. If the body is resistance to leptin and insulin, the pathway does not get excited and inhibited in the correct way which leads to more food intake (weight gain) and less energy usage.
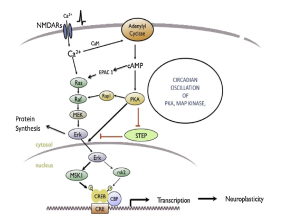
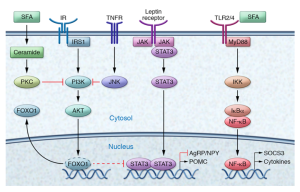 This image shows the many pathways involved in metabolic inflammation of the hypothalamus. This image just shows how complex inflammation is and how many moving parts there are. Inflammation of the hypothalamus affects many different cells types such as microglia, astrocytes, oligodendrocytes, and macrophages. Understanding the way that all the pieces fit together can help understand that obesity can be connected to the brain and help ways to properly treat obesity.
This image shows the many pathways involved in metabolic inflammation of the hypothalamus. This image just shows how complex inflammation is and how many moving parts there are. Inflammation of the hypothalamus affects many different cells types such as microglia, astrocytes, oligodendrocytes, and macrophages. Understanding the way that all the pieces fit together can help understand that obesity can be connected to the brain and help ways to properly treat obesity.
Proper Lifestyle Changes
 Proper weight loss is complex and there are many factors that play into it. Each persons body is different and needs different levels of nutrients so it is essential to not follow other peoples diets exactly. Looking at the food pyramid and creating balanced meals is the first step to changing a lifestyle. The focus should be on eating foods that make you feel good and not just cutting out foods.
Proper weight loss is complex and there are many factors that play into it. Each persons body is different and needs different levels of nutrients so it is essential to not follow other peoples diets exactly. Looking at the food pyramid and creating balanced meals is the first step to changing a lifestyle. The focus should be on eating foods that make you feel good and not just cutting out foods.
Physical activity is also important. Exercise of any kind can release certain chemicals in the brain to make you feel good. The focus should always be on what makes you feel the best. Knowing the lifestyles changes and knowing the science behind obesity can help people understand themselves more and seek out help from a trusted healthcare provider if they don’t see any changes. There are treatments options such as medication and surgeries that can help people if traditional eating and exercise changes do not work.
References:
en.wikipedia.org/wiki/Fad_diet
reidhealth.org/…/fad-diets-the-new-years-resolutions-worst-enemy
reidhealth.org/…/fad-diets-the-new-years-resolutions-worst-enemy





 pexels.com stock photo by Darya Sannikova
pexels.com stock photo by Darya Sannikova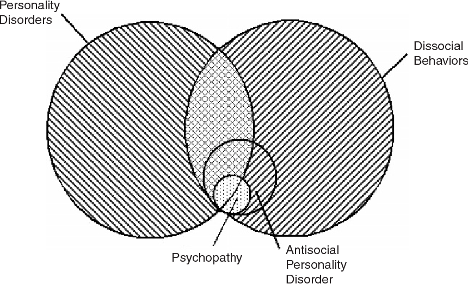



:max_bytes(150000):strip_icc()/what-is-a-psychopath-5025217-62ac72329fee4a16b4bad6f1980c421f.png)
:max_bytes(150000):strip_icc()/2795392-article-what-is-nature-versus-nurture-5a971887eb97de0036685ad3.png)

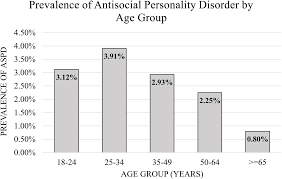
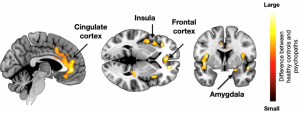
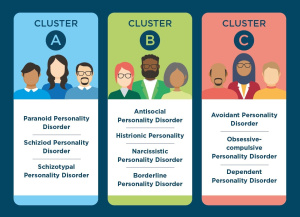
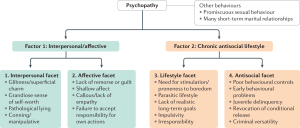 When diagnosing psychopathy, a questionnaire called the
When diagnosing psychopathy, a questionnaire called the 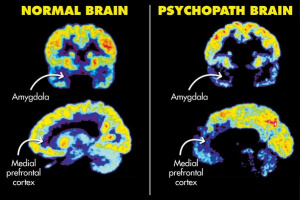 Researchers do not know much about the true cause of psychopathy and ASPD. Previous research has shown that there may be some
Researchers do not know much about the true cause of psychopathy and ASPD. Previous research has shown that there may be some  There are many stereotypes when it comes to the term psychopath. Most of these stereotypes come from media. Most notably, Joe Goldberg from the hit TV show You and Norman Bates from the hit TV show Bates Motel. The TV and movie industry has made a lot of money off of dramatizing people and the term psychopath, but these are not an accurate depiction of what it is like to live with the psychopath label (Norman and Norma Bates on the left and Joe Goldberg below).
There are many stereotypes when it comes to the term psychopath. Most of these stereotypes come from media. Most notably, Joe Goldberg from the hit TV show You and Norman Bates from the hit TV show Bates Motel. The TV and movie industry has made a lot of money off of dramatizing people and the term psychopath, but these are not an accurate depiction of what it is like to live with the psychopath label (Norman and Norma Bates on the left and Joe Goldberg below).
 Artstract #2
Artstract #2