Artstract by H. Puppe
Referred to as “the gate-way drug,” weed, Mary Jane, or pot, cannabis has been known by many for a long time. But many do not know how it works in the body. Cannabis acts on the endocannabinoid system. There are two main things to understand about the endocannabinoid system: endocannabinoids and CB1 and CB2 receptors.
Endocannabinoids and Receptors
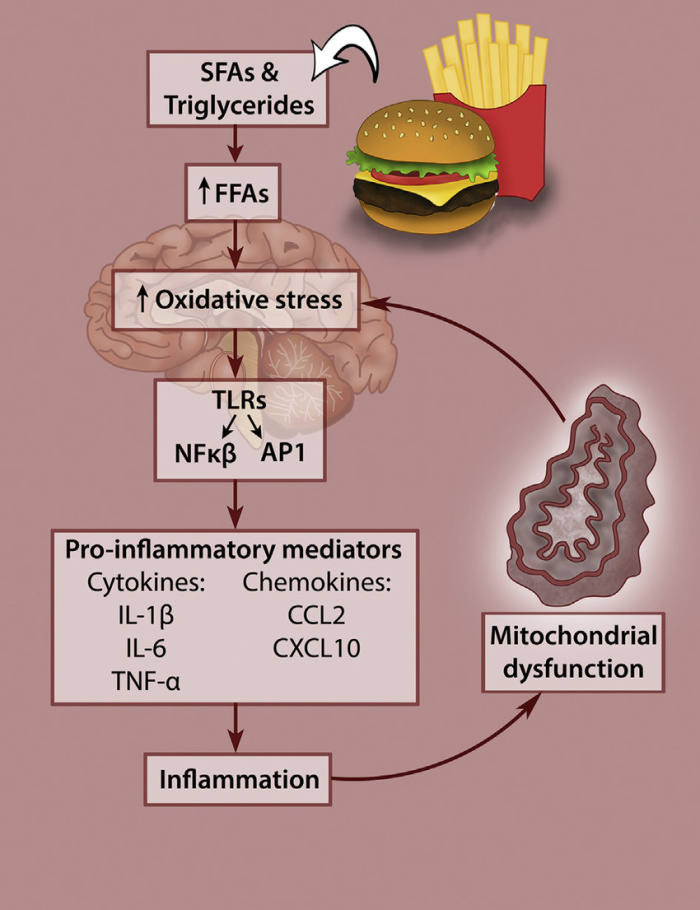
Endocannabinoids are chemical compounds that are produced in the body that bind to the same receptors as chemicals in cannabis like THC, which is the chemical that produces the euphoric effects. Two endocannabinoids are arachidonoylethanolamide (AEA) and 2-arachidonylglycerol (2-AG). Both of these act on CB1 and CB2 receptors. CB1 receptors are in the neocortex, hippocampus, basal ganglia, cerebellum, and brainstem as well as the testis, eyes, spleen, and the inner lining of veins, arteries, and capillaries. CB2 receptors are associated with inflammation and immune cells.1 Figure 1 shows the locations of CB1 and CB2 receptors in the body. Thus, it is safe to say that THC and endocannabinoids effect almost everything in the human body.
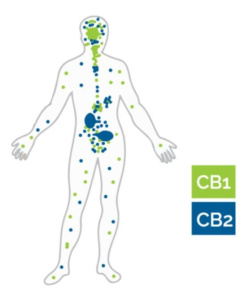
Figure 1. Locations of CB1 and CB2 receptors in the body
Treatment with THC
Legalizing THC has been a topic of debate for a long time, especially for medical use. THC has been used to help manage pain, nausea, weight and appetite, glaucoma, multiple sclerosis, Parkinson’s, and the list continues to grow. But, there is one major problem. Cannabis continues to be source of many problems like increased risk of car accidents, injury, and cannabis use disorder.2
Signs of cannabis use disorder include:
- Using more marijuana than intended
- Trying but failing to quit using marijuana
- Spending a lot of time using marijuana
- Craving marijuana
- Using marijuana even though it causes problems at home, school, work
- Despite social and relationship problems
- Despite physical or psychological problems
- Giving up important activities with friends and family in favor of using marijuana.
- Using marijuana in high-risk situations, such as while driving a car.
- Needing to use more marijuana to get the same high.
- Experiencing withdrawal symptoms when stopping marijuana use.
People who use cannabis have a 10% likelihood of developing cannabis use disorder.3 In a study with THC treatment, glaucoma patients had to smoke six to eight doses each day to alleviate symptoms. This led to tolerance of the drug while reducing the beneficial effects. When stopping treatment, patients experienced withdrawal symptoms. It was concluded that these high daily doses resulted in cannabis use disorder.4 Cannabis may not be the cure-all that people think it to be and it is important to look into the negatives along with the positives of cannabis use. Because of this, alternative treatment must be taken into consideration before prescribing medical cannabis.
Stimulating the Endocannabinoid System
Low endocannabinoid levels have been linked to major depressive disorder, multiple sclerosis, and Parkinson’s. If we want to treat with cannabis without actually using cannabis, we can look to stimulating the endocannabinoid system. There are other ways besides cannabis to stimulate this system.
- Cold Temperatures
-
- Exposure to cold temperatures can increase the amount of endocannabinoids in the body as well as the density of CB1 receptors. An easy way to achieve this is to take cold showers.
- Coffee and Tea
-
- Consuming caffeine has been shown to enhance endocannabinoids and CB1 receptors. Tea specifically also has an anti-inflammatory effects.
- Exercise
-
- Exercise results in upregulation of CB1 receptors and increased CB1 receptor sensitivity5
Although cannabis has many benefits that effects many parts of the body, there are many negatives. It may be possible to mimic its effects through the activities listed above and may be able to help treat these ailments.
References
- Kendall, D. A., & Yudowski, G. A. (2017). Cannabinoid receptors in the central nervous system: their signaling and roles in disease. Frontiers in Cellular Neuroscience, 10. https://doi.org/10.3389/fncel.2016.00294
- (2019). Cannabis (Marijuana) and cannabinoids: What you need to know. National Center for Complementary and Integrated Health. https://www.nccih.nih.gov/health/cannabis-marijuana-and-cannabinoids-what-you-need-to-know#:~:text=Drugs%20containing%20cannabinoids%20may%20be,loss%20associated%20with%20HIV%2FAIDS.
- (2020). Addiction (Marijuana or cannabis use disorder). National Center for Injury Prevention and Control, Center for Disease Control and Prevention. https://www.cdc.gov/marijuana/health-effects/addiction.html#:~:text=Some%20people%20who%20use%20marijuana,marijuana%20have%20marijuana%20use%20disorder
- Sun, X., Xu, C. S., Chadha, N., Chen, A., & Liu, J. (2015). Marijuana for glaucoma: a recipe for disaster or treatment? The Yale Journal of Biology and Medicine, 88(3), 265–9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4553646/
- Fallis, J. (2023). 26 powerful ways to boost your endocannabinoid system. Optimal Living Dynamics. https://www.optimallivingdynamics.com/blog/how-to-stimulate-and-support-your-endocannabinoid-system