Featured image (Artstract) created by Cayley Borrud with ChatGPT
Glioblastoma (GBM) is the most aggressive and most common type of brain cancer that originates in astrocytes. Currently, there is no cure for glioblastoma but there are treatments that can slow growth and make a more comfortable life. The main symptom is headaches that are very frequent and worsening [2]. Unfortunately, by the time symptoms show up, the tumor may have likely already degraded healthy brain matter and started growing rapidly. These poses the question; how does glioblastoma grow?
Understanding the step-by-step development of glioblastoma is important because each stage of tumor growth reveals a potential point for treatment.
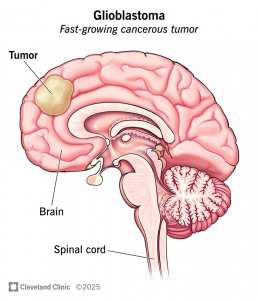 Image sourced from Cleveland Clinic
Image sourced from Cleveland Clinic
Tumor growth and development:
Glioblastoma can develop from one of two origins. The first way GBM can develop is very quick and develops within months without any pre-existing gliomas (Primary GBM). The second way it can develop is from a preexisting glioma (Secondary GBM). Secondary GBM develops very slowly over the course of years [2].
Despite the origins, glioblastomas arise from the same activations of pathways. The cancer would start out benign with its cAMP pathway being increased. The increase of this pathway is a good thing since it’s a suppressor of cancer cell growth [3]. Unfortunately, it can quickly become malignant where it mutates further and becomes aggressive. According to the article by Fung et al., in a malignant cancer, the cAMP pathway is suppressed while the MAPK and PI3K pathways become hyperactive [1]. The MAPK and PI3K pathways drive the cancer cell spread and growth.
While the GBM cells are multiplying, they are also producing enzymes called Matrix-metalloproteinases (MMPs) that break down the healthy brain tissue to make more room for it to spread [5]. Additionally, while the cancer cells are multiplying, they are creating new blood vessels that provide vital nutrients for the growth of the cancer. This process is called angiogenesis [6].
Then, the tumor progresses into a multiforme state. This means that the cells would develop different genetic traits. In Glioblastoma multiforme, the cells accumulate additional mutations and changes over time. This is how the cancer can become resistant to gene targeted therapies [4].
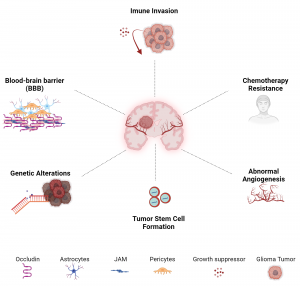 image sourced from biorender
image sourced from biorender
Takeaways:
Glioblastoma is not a single event but a multistep evolutionary process where cells mutate, invade, recruit blood supply and adapt to survive. Understanding each developmental step may help researchers design better therapies. In addition, the genetic diversity within GBM allows some cells to resist targeted therapies and continue progressing. Knowing how GBM invades healthy brain tissue can help scientists create drugs that block enzymes such as MMPs and slow tumor spread. Also, understanding how the tumor forms new blood vessels may improve anti-angiogenic therapies that cut off its nutrient supply.
As scientists continue to understand the molecular pathways behind GBM development, including MAPK, PI3K, and cAMP signaling, new treatment strategies may emerge. While glioblastoma remains devastating, advances in research offer hope for more effective and personalized therapies in the future.
Sources:
1. Fung, N. H., Grima, C. A., Widodo, S. S., Kaye, A. H., Whitehead, C. A., Stylli, S. S., & Mantamadiotis, T. (2019). Understanding and exploiting cell signalling convergence nodes and pathway cross-talk in malignant brain cancer. Cellular Signalling, 57, 2–9. https://doi.org/10.1016/j.cellsig.2019.01.011
2. Mayo Clinic Staff. (2026, March 5). Glioblastoma: Symptoms and causes. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/glioblastoma/symptoms-causes/syc-20569077
3. Xing, F., Luan, Y., Cai, J., Wu, S., Mai, J., Gu, J., Zhang, H., Li, K., Lin, Y., Xiao, X., Liang, J., Li, Y., Chen, W., Tan, Y., Sheng, L., Lu, B., Lu, W., Gao, M., Qiu, P., … Yan, G. (2017). The anti-Warburg effect elicited by the cAMP-PGC1α pathway drives differentiation of glioblastoma cells into astrocytes. Cell Reports, 18(2), 468–481. https://doi.org/10.1016/j.celrep.2016.12.037
4.Li, X., Wang, Y., Zhang, H., Chen, J., & Zhao, Q. (2025). Applications and emerging challenges of single-cell RNA sequencing technology in tumor drug discovery. Drug Discovery Today, 30(2), 104290. https://doi.org/10.1016/j.drudis.2025.104290
5. Aitchison, E. E., Dimesa, A. M., & Shoari, A. (2025). Matrix metalloproteinases in glioma: Drivers of invasion and therapeutic targets. BioTech, 14(2), 28. https://doi.org/10.3390/biotech14020028
6. Barresi, V., Christopoulou, M.-E., Karamani, V., Aletras, A. J., & Gatzounis, G. (2015). p-CREB expression in human meningiomas: Correlation with angiogenesis and recurrence risk. Journal of Neuro-Oncology, 122(1), 87–95. https://doi.org/10.1007/s11060-014-1706-9