Feature Image Credit: Created by Jackline Peace Nanyonga using ChatGPT.
Introduction: Why Gene Transcription Matters
Your brain is constantly interpreting signals and translating them into changes in gene expression. One of the most important pathways involved in this process is Wnt signaling, a highly conserved system that plays a central role in brain development and neural function.
What makes this pathway especially powerful is not just the signal itself, but how it ultimately influences gene transcription. At the center of this process is β-catenin, a protein that acts as the final decision-maker – determining whether specific genes are turned on or off.
Understanding how β-catenin regulates transcription provides insight into how disruptions in signaling pathways may contribute to neurodevelopmental disorders such as schizophrenia.
When the Pathway is Off: Controlling β-Catenin
In the absence of a Wnt signal, β-catenin is tightly regulated and continuously broken down. This is achieved through a “destruction complex” composed of proteins such as GSK3β, Axin, APC, and CK1α. This complex phosphorylates β-catenin, targeting it for degradation and preventing it from accumulating in the cell (Singh, 2013).[1]
As a result, β-catenin cannot enter the nucleus, and Wnt target genes remain inactive. In this state, the pathway is effectively turned off, and no transcriptional response occurs.
Turning the Switch On: β-Catenin and Gene Activation
When Wnt ligands bind to their receptors, the destruction complex is disrupted. This allows β-catenin to escape degradation and accumulate in the cytoplasm. As levels rise, β-catenin translocates into the nucleus, where it interacts with TCF/LEF transcription factors to activate gene expression programs involved in neural development, cell proliferation, and synaptic plasticity (Singh, 2013).[1]
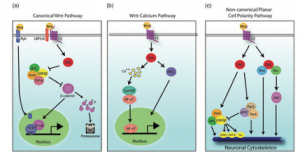
Figure 1: Canonical Wnt signaling pathway showing β-catenin degradation (left) versus nuclear translocation and transcriptional activation (right).[1]
Importantly, β-catenin does not bind DNA directly. Instead, it acts as a co-activator, helping transcription factors initiate gene expression. This makes it a crucial link between extracellular signals and long-term cellular changes.
Regulation and Relevance: Why β-Catenin Matters in the Brain
The activity of β-catenin is closely tied to GSK3β, a kinase that determines whether β-catenin is degraded or stabilized. When GSK3β is active, β-catenin is broken down. However, when GSK3β is inhibited – such as by lithium – β-catenin accumulates and transcription is enhanced (Singh, 2013).[1]
This regulation becomes especially important in the context of schizophrenia. The paper highlights that multiple pathways, including dopamine signaling and Akt/GSK3β signaling, converge on β-catenin. Additionally, genetic factors such as the DISC1 gene can inhibit GSK3β, leading to increased β-catenin stability and greater activation of transcription (Singh, 2013).[1]
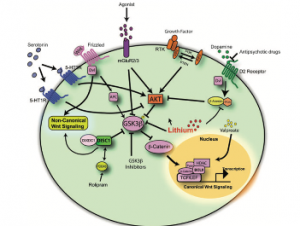 Figure 2 (left): Integration of dopamine, Akt/GSK3β, and Wnt signaling pathways converging on β-catenin-mediated transcription.[1]
Figure 2 (left): Integration of dopamine, Akt/GSK3β, and Wnt signaling pathways converging on β-catenin-mediated transcription.[1]
These findings suggest that β-catenin is not just part of one pathway, but a central hub where multiple signals influence gene expression.
Conclusion: A Small Protein with Big Consequences
β-catenin represents the point where signaling pathways are translated into changes in gene expression. By controlling TCF/LEF-mediated transcription, it plays a key role in shaping how neurons develop, connect, and function.
Because of this, even subtle disruptions in β-catenin regulation can have significant downstream effects. In disorders like schizophrenia, these disruptions may alter gene expression patterns during critical stages of brain development, contributing to long-term changes in neural circuitry.
Ultimately, studying β-catenin helps shift our understanding of psychiatric disorders—from focusing only on neurotransmitters to considering how gene transcription itself is regulated.
Bibliography
[1]
K. K. Singh, “An emerging role for Wnt and GSK3 signaling pathways in schizophrenia,” Clin Genet, vol. 83, no. 6, pp. 511–517, Jun. 2013, doi: 10.1111/cge.12111.
