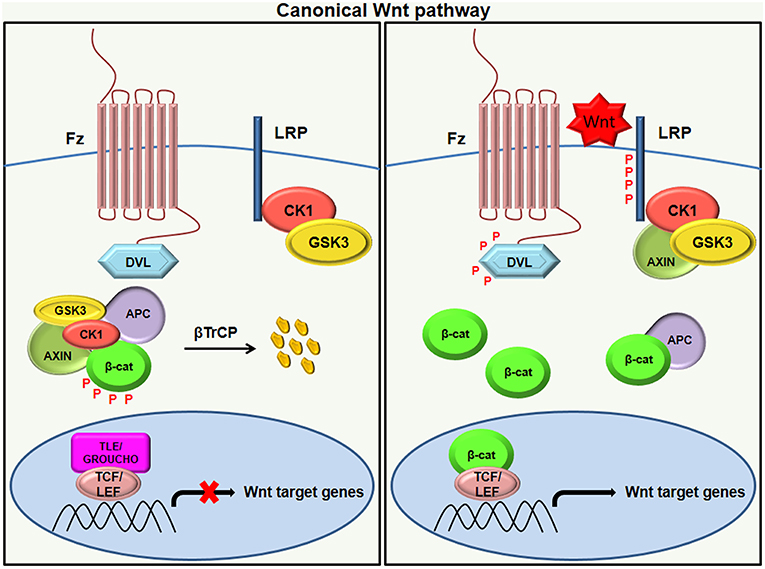
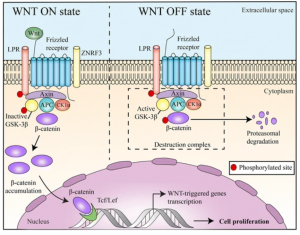
Addiction on the Molecular Level

Addiction is a significant issue in today’s society. Addiction is defined as a compulsive and persistent dependence on a substance or behavior that has negative consequences. Some of the most common types of addiction are substance addiction (addiction to drugs, alcohol, and tobacco), natural addiction (addiction to food, binge eating disorder) and behavioral addiction (addiction to gambling, video games, and social media).
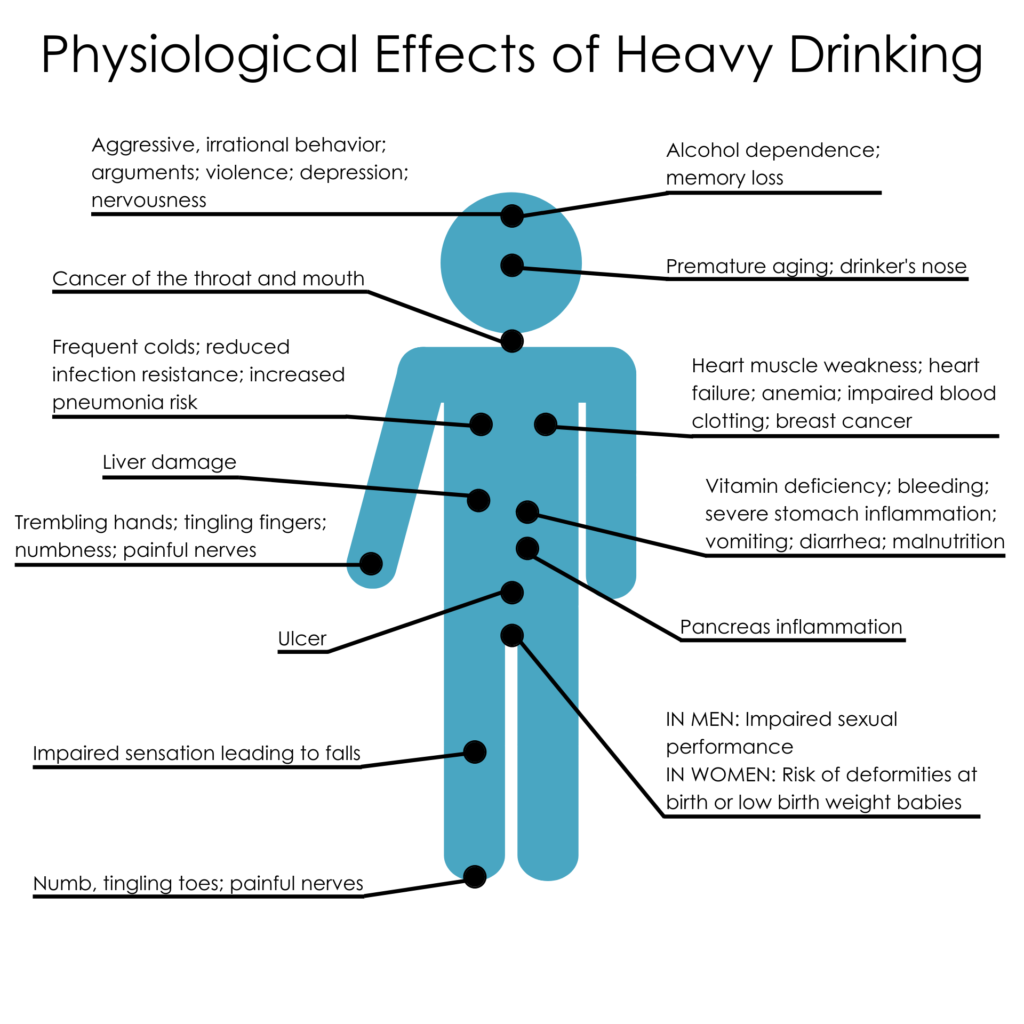
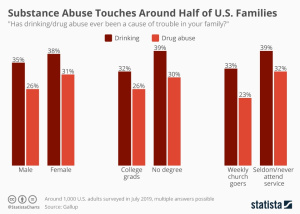
Addiction can have a significant impact on an individual’s life as well as the lives of those close to them. It can result in physical and mental health issues, financial difficulties, social isolation, and legal complications. Addiction can also have a negative impact on the larger community by causing crime, violence, and other social problems.
Addiction is particularly problematic in today’s society due to a number of factors. For starters, addictive substances and behaviors are more widely available, thanks to the proliferation of online gaming and social media platforms. Second, there is less social stigma attached to addiction, which may make it easier for people to engage in addictive behaviors without fear of being judged. Finally, many people lack access to effective addiction treatment and support services, making it difficult for them to overcome their addiction and live a healthy life.
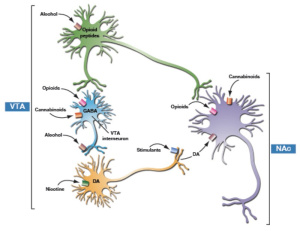
Drug abuse can activate several signaling pathways in the brain, leading to the development of addiction. The following are some of the most common signaling pathways involved in drug abuse:
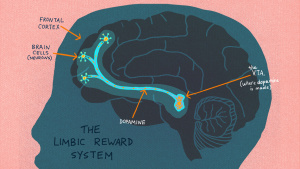
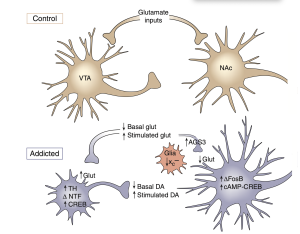
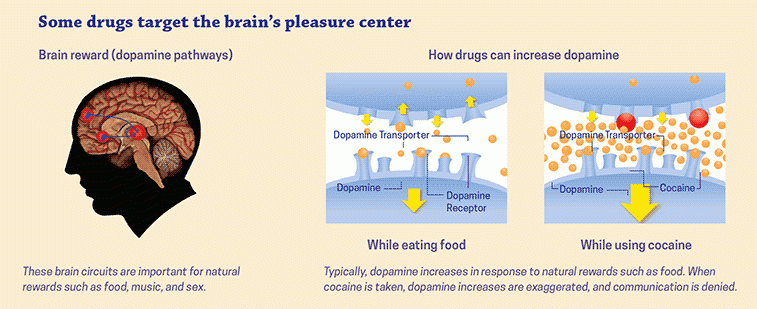
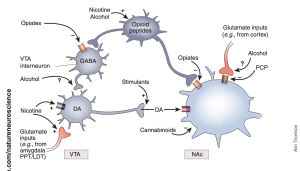
- Dopamine pathway: The mesolimbic dopamine pathway, which is a circuit of brain regions involved in reward and motivation, is one of the key pathways identified by Dr. Nestler and his colleagues. This pathway is activated when a person engages in rewarding behavior or takes an addictive drug, and dopamine is released in the nucleus accumbens, a key region of the brain’s reward circuitry. This dopamine release reinforces and motivates the individual to repeat the behavior.
- Glutamate pathway: The glutamate pathway is responsible for learning and memory. Drugs of abuse can activate this pathway, leading to changes in synaptic plasticity, which can lead to drug cravings and addiction.
- GABA pathway: The gamma-aminobutyric acid (GABA) pathway is responsible for the brain’s inhibitory system. Drugs of abuse can disrupt the balance of GABA signaling, leading to an increase in excitatory signaling, which can lead to drug cravings and addiction.
- Endocannabinoid pathway: The endocannabinoid system is responsible for regulating appetite, pain, and mood. Drugs of abuse can activate the endocannabinoid pathway, leading to changes in appetite and mood.
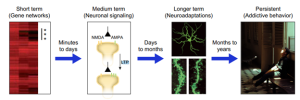
Moreover, repeated drug use or addictive behavior causes the brain to adapt, resulting in long-term changes in gene expression and synaptic plasticity. Dr. Nestler has identified a number of specific molecular pathways involved in these adaptations, such as changes in gene expression of various transcription factors, epigenetic DNA modifications, and changes in synaptic plasticity and neuroplasticity.
While there may not be a single common molecular pathway for addiction, Dr. Nestler’s research has contributed to the identification of some of the key molecular and cellular mechanisms that underpin addictive behavior. This understanding is critical for the development of new treatments and interventions to assist individuals in overcoming addiction and leading healthy, fulfilling lives.
sources:
https://moodle.cord.edu/pluginfile.php/1266921/mod_resource/content/4/Nestler%202005.pdf
https://www.ncbi.nlm.nih.gov/books/NBK424849/figure/ch2.f5/








 Statistically,
Statistically, 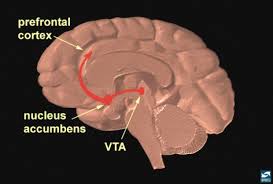 Addicts will choose this feeling over anything else in their life because eventually their brains don’t feel motivation or reward for any other activity other than using their drug of choice. Even with their drug of choice, they will eventually need to increase the amount they take to keep that high.
Addicts will choose this feeling over anything else in their life because eventually their brains don’t feel motivation or reward for any other activity other than using their drug of choice. Even with their drug of choice, they will eventually need to increase the amount they take to keep that high.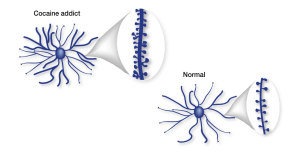 Part of the brain responsible for reason, judgment, learning, and memory begin to have changes as well. In some spots, the neurons are pruned back whereas in other regions there are more connections, completely changing the shape of the neuron. After all of these changes, the drug-seeking behavior becomes habit and the person becomes addicted.
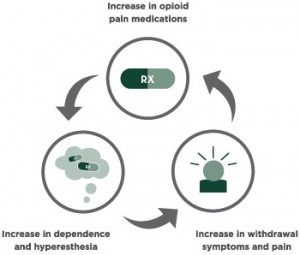
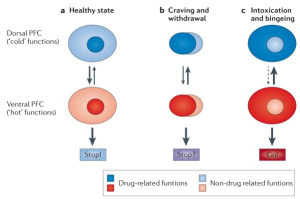
Part of the brain responsible for reason, judgment, learning, and memory begin to have changes as well. In some spots, the neurons are pruned back whereas in other regions there are more connections, completely changing the shape of the neuron. After all of these changes, the drug-seeking behavior becomes habit and the person becomes addicted. Relapse is defined as when a person returns to using drugs or alcohol after a period of sobriety. There are many factors that contribute to relapsing, which can be seen in this image. After the neurons are changed and the feeling and memories of drugs are implanted into someones brain it can be extremely hard to just give it up. The brain is still searching for this increased reward feeling and it isn’t getting it from ‘normal’ daily activities which can make turning back to drugs really tempting.
Relapse is defined as when a person returns to using drugs or alcohol after a period of sobriety. There are many factors that contribute to relapsing, which can be seen in this image. After the neurons are changed and the feeling and memories of drugs are implanted into someones brain it can be extremely hard to just give it up. The brain is still searching for this increased reward feeling and it isn’t getting it from ‘normal’ daily activities which can make turning back to drugs really tempting.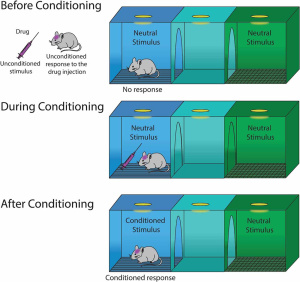 Conditioning is defined as a higher dose of drugs that can be tolerated when external stimuli is associated with prior administration of the drug is present. Basically, this means that if someone is doing drugs in the same room at the same time everyday they will eventually build up a tolerance because their brain will release the the effects of the drug before it is even taken. This leads to a tolerance, and if the drug is taken with any of the external factors changed it can lead to an overdose.
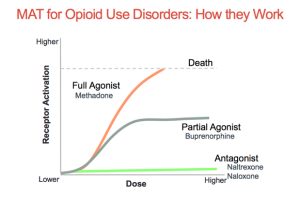
Conditioning is defined as a higher dose of drugs that can be tolerated when external stimuli is associated with prior administration of the drug is present. Basically, this means that if someone is doing drugs in the same room at the same time everyday they will eventually build up a tolerance because their brain will release the the effects of the drug before it is even taken. This leads to a tolerance, and if the drug is taken with any of the external factors changed it can lead to an overdose.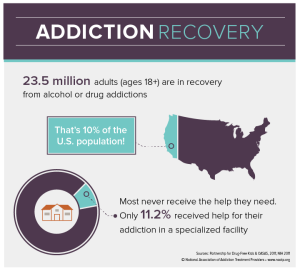 Recovery is much more complex than it is made out to be. There are many factors that play a part in the life of an addict. It is not as simple as quitting cold turkey sometimes. There are memories and outside factors that can effect the ability to remain sober. Being educated on all the facts of addiction can help stop the stigma that it is just a simple choice. It truly goes down to the cellular and molecular level and changes are happening at those levels making it difficult to change behaviors right away. Treatment for addiction can be very expensive and difficult.
Recovery is much more complex than it is made out to be. There are many factors that play a part in the life of an addict. It is not as simple as quitting cold turkey sometimes. There are memories and outside factors that can effect the ability to remain sober. Being educated on all the facts of addiction can help stop the stigma that it is just a simple choice. It truly goes down to the cellular and molecular level and changes are happening at those levels making it difficult to change behaviors right away. Treatment for addiction can be very expensive and difficult.