Feature Image: Artstract by Emma Doty using AI
Overview:
Psychostimulant use disorder (PUD) is a chronic, relapsing condition characterized by compulsive drug use despite harmful consequences. The review article (access here) emphasizes that addiction is not simply a behavioral issue, but a disorder rooted in long-lasting changes in brain circuitry. Specifically, the paper highlights how psychostimulants such as cocaine, amphetamine, and nicotine alter glutamatergic signaling within the brain’s reward system.[1]
Role of Glutamate:
At the center of this process is glutamate, the brain’s primary excitatory neurotransmitter. The article explains that psychostimulant use disrupts normal glutamate transmission and receptor function, particularly involving the metabotropic glutamate receptors (mGluRs). These disruptions lead to neuroadaptations that alter synaptic plasticity and reinforce drug-seeking behaviors. These changes remain even without the presence of the drug which contributes to relapses.[2]
Learning and Memory Impact:
Importantly, the paper frames addiction as a form of maladaptive learning, as glutamate is essential in learning and memory processes. Drug-related experiences become ingrained in the brain’s reward circuitry, including regions like the nucleus accumbens, prefrontal cortex, and amygdala. Strong memories are formed that associate the stimulant with a feeling of pleasure which rewires the brain to seek these drugs. Over time, these changes alter how individuals respond to rewards, stress, and environmental cues, making it increasingly difficult to break the cycle of addiction.[3]
Long-Term Potentiation:
To fully understand how these long-lasting changes occur, it is essential to consider long-term potentiation (LTP). LTP refers to the persistent strengthening of synapses following a pattern of intense activity and is widely regarded as the cellular basis of learning and memory.[4]
This intense neuronal activity leads to the release of large amounts of glutamate, which signifies the beginning of this process. Glutamate then activates AMPA receptors, causing depolarization of the postsynaptic neuron. When this depolarization is strong enough, it removes the magnesium block from NMDA receptors, allowing a rapid influx of calcium. This acts as a molecular trigger, activating proteins such as CaMKII, which promote the insertion of additional AMPA receptors into the postsynaptic membrane. These extra receptors are inherently more sensitive to glutamate. As a result, the synapse becomes more responsive to future signals, strengthening the connection between neurons.[5]
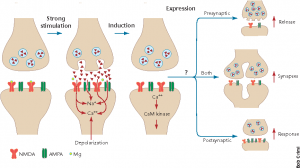
Image of LTP Mechanism: https://www.researchgate.net/figure/nduction-and-expression-of-LTPStrong-repetitive-stimulation-of-the-input-to-a_fig2_12895506
LTP occurs in two phases: an early phase (E-LTP), which occurs independently of protein synthesis, and a late phase (L-LTP), which requires gene expression and structural changes to the synapse. During this late phase, transcription factors like CREB drive the growth of dendritic spines and the expansion of synaptic contact area, allowing these strengthened connections to persist for hours, days, or even longer.[6]
Final Thoughts:
In the context of addiction, LTP provides an explanation for the role of learning and memory. Psychostimulants effectively hijack the brain’s natural learning processes by inducing LTP within reward circuits. Each drug exposure strengthens the neural pathways associated with drug use, making those pathways more easily activated in the future.
Ultimately, the article emphasizes that targeting glutamatergic signaling and synaptic plasticity, such as the LTP mechanism, may be key to developing effective treatments for addiction. By disrupting or reshaping these maladaptive neural connections, it may be possible to weaken the hold that drug-related memories have on behavior.
[1] Mozafari et al., “A Review on the Role of Metabotropic Glutamate Receptors in Neuroplasticity Following Psychostimulant Use Disorder,” Progress in Neuro-Psychopharmacology and Biological Psychiatry 124 (June 2023): 110735, https://doi.org/10.1016/j.pnpbp.2023.110735.
[2] Mozafari et al., “A Review on the Role of Metabotropic Glutamate Receptors in Neuroplasticity Following Psychostimulant Use Disorder.”
[3] Mozafari et al., “A Review on the Role of Metabotropic Glutamate Receptors in Neuroplasticity Following Psychostimulant Use Disorder.”
[4] “What Is Long-Term Potentiation and How Does It Work?,” ScienceInsights, November 21, 2025, https://scienceinsights.org/what-is-long-term-potentiation-and-how-does-it-work/.
[5] ScienceInsights, “What Is Long-Term Potentiation and How Does It Work?”
[6] “Long-Term Potentiation – an Overview | ScienceDirect Topics,” accessed March 24, 2026, https://www.sciencedirect.com/topics/neuroscience/long-term-potentiation.
