Featured image created by Julia Wolf and Microsoft CoPilot
The Science of Addiction
Addiction is no longer understood as simply a behavioral problem, it is a disorder rooted in long-lasting changes in the brain’s rewiring. Psychostimulant drugs like cocaine, amphetamine, and nicotine target the brain’s reward system, a network that includes the ventral tegmental area (VTA), nucleus accumbens (NAc), prefrontal cortex (PFC), amygdala, and hippocampus. These regions normally work together to guide behavior towards rewarding experiences like food, social interaction, and survival. Repeated drug exposure hijacks this system, strengthening pathways associated with the drug and making drug-seeking behavior automatic and persistent.
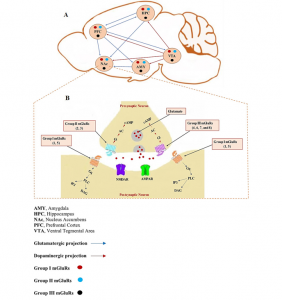
A major player in these changes is the family of metabotropic glutamate receptors (mGluRs). These G-protein coupled receptors regulate longer-term, slower changes in synaptic strength. In particular, Group 1 mGluRs (mGluR1 and mGluR5) influence extracellular calcium levels, enzyme activity, and downstream signaling pathways, shaping learning and memory processes. Activation of these receptors can reinforce drug-seeking behavior, while blocking them can reduce craving and relapse. These interactions show that mGluRs have a central role in addiction-related neuroplasticity.
To learn more about the science of addiction, click here.
Long-Term Depression (LTD): The Brain’s Way of Letting Go
Long-term depression, or LTD, is the brain’s way of intentionally weakening synaptic connections. While long-term potentiation (LTP) strengthens certain pathways, LTD removes or diminishes connections that are no longer useful, helping the brain maintain balance and adapt to new information. LTD is essential for fine-tuning neural networks, pruning irrelevant memories, and preventing overexcitation that could damage neurons [2].
LTD occurs at excitatory synapses in key brain areas such as the hippocampus, cortex, striatum, and cerebellum. In the hippocampus, LTD helps clear out old or irrelevant memories, ensuring that new learning isn’t cluttered by outdated information. In the cerebellum, it fine-tunes motor coordination, helping us master smooth, precise movements. It also happens in other regions like the neocortex, striatum, and amygdala, where it contributes to emotion regulation, decision-making, and habit formation [4].
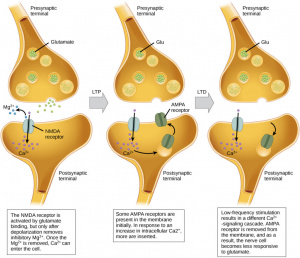
There are different forms of LTD. Homosynaptic LTD weakens the synapse that is directly activated, while heterosynaptic LTD can weaken neighboring synapses that weren’t directly stimulated, helping the brain maintain balance across its networks [5]. At the molecular level, most LTD mechanisms involve removing AMPA receptors from the postsynaptic membrane, which reduces the strength of the synapse. In NMDA receptor-dependent LTD, common in the hippocampus, low-frequency signals from presynaptic neurons allow a small influx of calcium into the postsynaptic neuron, which activates enzymes that pull AMPA receptors out of the membrane. This results in a long-term decrease in synaptic strength, helping the brain fine-tune connections after individual events [6].
Another form, mGluR-dependent LTD, is found in both the hippocampus and cerebellum. Here, metabotropic glutamate receptors detect glutamate and trigger a cascade of signals inside the neuron, eventually removing AMPA receptors. This process is slower than NMDA-LTD but is important for adapting to prolonged experiences, such as repeated learning tasks or chronic drug exposure [7].
LTD isn’t just about pruning; it is very connected to memory, learning, and addiction. By eliminating unnecessary synapses, LTD supports efficient memory storage and refines motor skills. Disruption of mGluR-dependent LTD in reward pathways can make drug-related cues overly strong, contributing to addiction. Studying LTD helps us understand how the brain balances strengthening important connections while removing the ones we don’t need, offering insights into memory, motor learning, and potential therapies for addiction and neurological disorders [4].
To learn more about LTD functions and mechanisms, watch this short video!
Why This Matters: Addiction as a Failure to Adapt
Addiction can be seen as a form of maladaptive learning, a problem that is increasingly relevant in today’s society, not just with drugs, but also with behaviors like excessive phone use, social media, and gaming. The brain’s natural ability to adapt, strengthening useful pathways while weakening those that aren’t needed, can be redirected in harmful ways. Circuits linked to addictive behaviors become stronger and more dominant, while the brain struggles to weaken or “forget” behaviors that are harmful.
Studying mechanisms like long-term depression (LTD) and the role of mGluRs helps explain why cravings are so persistent and why relapse is common. This scientific understanding also highlights potential paths for treatment: therapies that restore the brain’s ability to weaken harmful connections or rebalance glutamate signaling may help people regain control over their behavior. Addiction isn’t just about desire; it’s about the brain being caught in a pattern it can’t easily reset and understanding this can guide more effective interventions.
References
[1] R. Mozafari, S. Karimi-Haghighi, M. Fattahi, P. Kalivas, and A. Haghparast, “A review on the role of metabotropic glutamate receptors in neuroplasticity following psychostimulant use disorder,” Progress in Neuro-Psychopharmacology and Biological Psychiatry, vol. 124, p. 110735, Jun. 2023, doi: https://doi.org/10.1016/j.pnpbp.2023.110735.
[2] D. Purves et al., “Long-Term Synaptic Depression,” Neuroscience. 2nd edition, 2001, Available: https://www.ncbi.nlm.nih.gov/books/NBK10899/
[3] Lumen Learning, “Synaptic Plasticity | Biology for Majors II,” Lumenlearning.com, 2008. https://courses.lumenlearning.com/wm-biology2/chapter/synaptic-plasticity/
[4] C. Lüscher and K. M. Huber, “Group 1 mGluR-Dependent Synaptic Long-Term Depression: Mechanisms and Implications for Circuitry and Disease,” Neuron, vol. 65, no. 4, pp. 445–459, Feb. 2010, doi: https://doi.org/10.1016/j.neuron.2010.01.016.
[5] Z. I. Mannan, S. Azam, R. K. Budhathoki, M. Nur Alam, and H. Kim, “Modeling homosynaptic and heterosynaptic plasticity with a single neuromemristive synapse,” Journal of Advanced Research, Oct. 2025, doi: https://doi.org/10.1016/j.jare.2025.10.031.
[6] D. Saal and R. C. Malenka, “The role of synaptic plasticity in addiction,” Clinical Neuroscience Research, vol. 5, no. 2–4, pp. 141–146, Nov. 2005, doi: https://doi.org/10.1016/j.cnr.2005.08.009.
[7] S. J. Kang and B.-K. Kaang, “Metabotropic glutamate receptor dependent long-term depression in the cortex,” The Korean Journal of Physiology & Pharmacology, vol. 20, no. 6, p. 557, 2016, doi: https://doi.org/10.4196/kjpp.2016.20.6.557.