
Out of the various brain-related afflictions discussed in these posts, addiction is unique in that it doesn’t arise from faulty wiring, but rather from something external. This is primarily why it might not be as charitably framed in our society. Addiction is more often seen as a moral failing, a choice — something that willpower alone should fix. Those framings have real consequences: they shape how families respond to loved ones in crisis, how courts sentence people who relapse, and how little urgency surrounds the search for effective treatments. For psychostimulants — cocaine, amphetamine, methamphetamine, nicotine — there are still no FDA-approved medications.
The scientific picture tells a different story. Addiction is a chronic, relapsing disorder driven by lasting changes in brain circuitry: changes that affect memory, motivation, impulse control, and the receptors that regulate how neurons communicate. Most of those changes don’t reverse when someone stops using. Some get worse. The assumption that the longer someone stays clean, the safer they are from relapse turns out to be wrong in important ways.
A 2023 review published in Progress in Neuropsychopharmacology & Biological Psychiatry examined how a family of receptors called metabotropic glutamate receptors (mGluRs) reshape the reward circuit during and after psychostimulant use. [1] What emerges is a picture of a brain that doesn’t simply reset when drug use stops; instead, it keeps changing, often in ways that increase vulnerability to relapse.
The receptor system nobody talks about
Most public conversation about addiction focuses on dopamine, but glutamate (the brain’s primary excitatory neurotransmitter) is equally central to why addiction is so hard to escape. Glutamatergic circuits connect the prefrontal cortex, the nucleus accumbens (NAc), the amygdala, and the ventral tegmental area: the core of the brain’s reward system. The mGluRs that sit throughout this circuit act as volume knobs, tuning the strength of signals flowing between these regions.
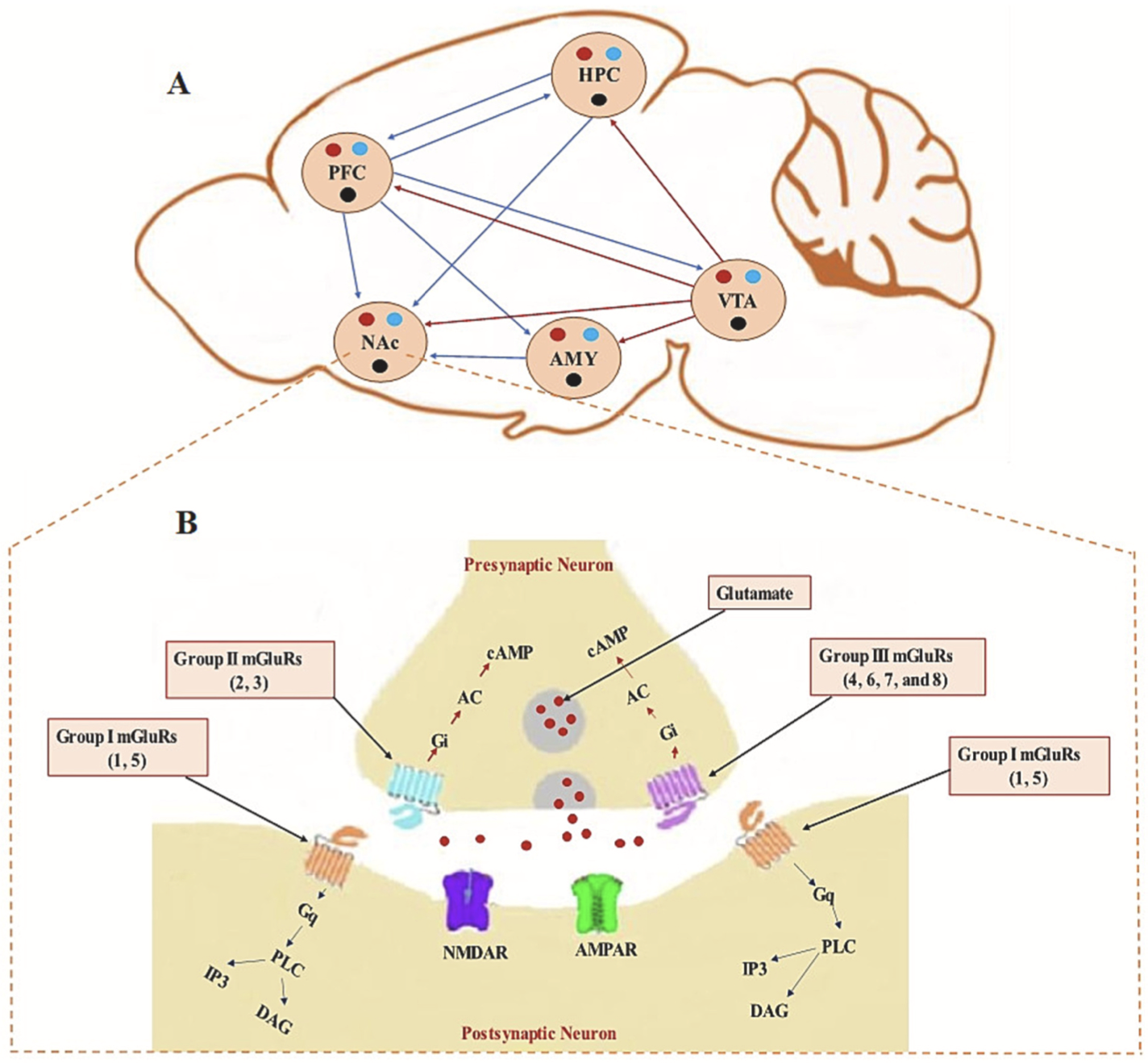
The review identifies three groups of mGluRs, each playing a distinct role. Group I receptors (mGluR1 and mGluR5), found mostly on the postsynaptic side, drive forms of synaptic strengthening and weakening that underlie learning and memory. Group II and III receptors sit primarily on presynaptic terminals and act as brakes, reducing glutamate release (Figure 1). [1] Psychostimulants like the drugs described in the first paragraph disrupt all of them.
What abstinence looks like
Consider someone who has been clean for two months. By most accounts, they are doing everything right and are on the road to recovery. The biology suggests, however, that they may actually be at greater risk of relapse than they were the week they quit. This phenomenon, the reason why the risk of relapse increases with time, is called the incubation of craving and was first documented in 1986. [2] Since then, it has been confirmed across cocaine, methamphetamine, heroin, alcohol, and nicotine.
The mGluR research helps explain why. During extended withdrawal, calcium-permeable AMPA receptors accumulate in NAc synapses, hardwiring drug-associated memories deeper into the circuit. [3] This is partly driven by a decline in mGluR1 activity. When researchers restore mGluR1 function, that accumulation reverses and incubated craving decreases, suggesting mGluR1 normally acts as a brake that gradually fails during abstinence. [4] At the same time, the prefrontal cortex remains persistently dysregulated in imaging studies of people in protracted withdrawal, undermining the impulse control needed to resist when a cue appears.
What this means
The review is careful to frame mGluRs as potential therapeutic targets precisely because the current situation is untenable: there are no FDA-approved medications for psychostimulant use disorder. The mGluR system offers multiple intervention points, from dampening cue reactivity to restoring glutamate homeostasis to interrupting the synaptic changes that incubate craving. But the broader implication is a clinical and cultural one. Telling someone that time and willpower are sufficient is not just incomplete — it runs counter to the neuroscience. The brain changes that drive relapse are not signs of weakness or poor motivation; they are the predictable outcome of a system that was, at the molecular level, systematically rewritten. Recovery is not as simple as merely abstaining, and understanding why is the first step toward correction.