As my time at Concordia comes to a close, I can’t help but reflect on the past four years and be truly grateful for the experience I have had. I became a part of a family like no other where talking about corn was “cool” and knowing people around every corner you turned was the norm. I had the ability to become close with my professors and build life long relationships.
Looking at neurochemistry as my capstone course, I have learned in a new way that I hadn’t experienced until this class. This course opened my eyes even more than they already were to the equality between students and professors. I was able to fully apply what I learned in this course to my outside relationships and life as a whole. My friends and family found it very interesting when I would come back from class every day and describe various neurological disorders and their causes because the information was so applicable.
I truly believe my education at Concordia has fully provided me with a fantastic liberal arts education and that “BREW” (Becoming Responsibly Engaged in the World) has been instilled in me.
The five goals of liberal arts learning at Concordia are: 1. Instill a love for learning 2. Develop foundational skills and transferable intellectual capacities 3. Develop an understanding of disciplinary, interdisciplinary, and intercultural perspectives and their connections 4. Cultivate an examined cultural, ethical, physical and spiritual self-understanding 5. Encourage responsible participation in the world
Neurochemistry has fully enveloped each of these five goals.
Every class period, as I mentioned previously, I would immediately tell a friend or family member about what I had learned and constantly desired to know more and gain a better understanding of each topic. Another aspect of this course I enjoyed was being able to apply what I had learned in neurochemistry to the anatomy course to accumulate more understanding and hear about other factors that were not discussed in the other course. As a whole, it allowed me to put the puzzle pieces together and make connections to obtain more knowledge on the subject area.
If I had the choice to do college all over, there isn’t anything I would change about it. Now that I am wanting to move onto dental school, I believe that I have an advantage by having the cultural awareness and the course work from every major. These classes have enhanced my learning by broadening my knowledge, thinking in different ways, and above all, finding an appreciation for every individual’s talents and not putting a hierarchy on anyone. But most importantly, I have learned how to become responsibly engaged in the world, not because thats what I’ve been told to do, but rather, because it is what I want to do.
Hats off to You, Neurochem!
Concordia College’s capstone Neurochemistry class taught by Dr. Julie Mach is the apex of a liberal arts education for a science major. It is both a class that pushes students outside of their comfort zone and a class that warrants great fun to be had. In this class, myself, my thirteen classmates, and Dr. Mach explored research about the brain’s mechanisms in disorders including obesity, Alzheimer’s, bulimia, Parkinson’s, schizophrenia, autism, and concussions. The structure of this class was far from a traditional lecture/assessment setting and unlike anything I have ever experienced before. Basically, an outsider could call us some brain nerds reading papers written by brain nerds with their PhD’s. In order to express my love for the class and reflect on how the class changed me as a student. I’ll explain an average week in the course.
Monday:
For me, Mondays were the most difficult day by far. The mission of the previous weekend was to read and analyze a 8-25 page paper on that week’s neurological topic. This sounds simple enough, right? Wrong. These papers were dense and complex yet often revolutionary. I would come to class feeling like I was completely lost in the paper yet in the back of my head had a feeling that these findings have huge implications in the science, medical, and social words. When we met as a class, we would spend the day discussing what we felt was important in the paper. What I loved about Monday’s was that we all were in the same boat: lost and looking for answers. We had the opportunity to sit in a circle, share our interpretations, and extrapolate the key concepts on the papers. As the day ended, we narrowed down our focus to 14 topics that we wanted to further research because we felt that they were critical in our understanding of the paper. Usually, as a type-A science major, the lack of understanding that accompanied Monday’s would send me into a panic. For this class, Monday’s were calming because I knew that this lack of understanding was a place to grow and that Dr. Mach would guide us in the right direction.
Wednesday:
Wednesdays were a day for everyone to live in the limelight. Between Monday and Wednesday, we all would research one specific topic related to the paper we are focusing on for the week and Dr. Mach would arrange “speed dating”. During speed dating, we would get four minutes with each other student were we could share what we personally researched to strengthen the entire class’s understanding on the neurological disorder. By the end, we had learned 14 new pieces of information related to the neurochemistry behind the diseases. No matter a person’s background or personality, everyone could positively contribute to the conversations. I always left Wednesday feeling full of knowledge and an understanding of the research I had feared just two days before.
Friday:
Fridays in Neurochemistry were my favorite days of the class. We would move across campus to some comfy chairs and discuss. Rather than discussing purely the science behind these neurological disorders, we had the reign to discuss every aspect of them. This includes treatments, ethics, and social factors. It’s easy to be scientists studying the brain and what’s malfunctioning in it. But to truly help the population, I think it’s extremely important to examine the entire person and the life that they live. On Fridays, we spent hours having meaningful and deep conversations about neurological disorders that affect millions of people each year. Even if we disagreed on a topic, the atmosphere of the class was one that encouraged resolution of conflict and understanding. My favorite time of my school weeks were Friday afternoons where I could curl up in a chair with a cup of coffee and learn from my brilliant classmates and hear their opinions.
One would think that lecture style classrooms would be the most effective when learning about something as objective as the brain. After taking this neurochemistry course, I would disagree. I learned more about the brain and its disorders in this class than my entire career as a Neurochemisty major at Concordia. I owe this to my professor and my classmates. Without Dr. Mach’s innovation and atypical approach to the class, I my mind would have been stuck in the walls of a PowerPoint lecture. Without my classmates, my own knowledge wouldn’t have been tested and I wouldn’t have gained knowledge from a wide range of experiences.
To Concordia, to Dr. Mach, to my classmates, I say thank you. Thank you for pushing my mental limits and thank you for all of the new knowledge on my favorite organ: the brain.
Don't Let FOMO Get The Best Of You
How to sum up my semester of Neurochemistry? I’m not exactly sure… There needs to be more research done… My understanding of it is…
This class was unlike any other that I’ve taken so far. Being a science major you mostly get to learn what other people have already discovered. Theories and laws that have been researched and proven for years. Even in lab I do experiments that I already know what I should be getting.
Neuroscience is a very new field. There is a lot of knowledge about it, but there is so much more that we don’t know. Many of the papers we read and theories we looked at were very recent. A lot of times we found ourselves at a roadblock where we really couldn’t find any more information because it wasn’t out there. There wasn’t enough research on the topic to solidify any facts. There could be a lot of theories or potential, but wasn’t proven yet.
That was another cool part of all the research that we did, we got to hypothesize our own ideas. We could look at a broad spectrum of possibilities and make our own connections. Were they always correct? Maybe not. The words, “My understanding” or “I think” were never frowned upon in this class, they were encouraged. If the best scientists in the world couldn’t figure it out exactly, then why couldn’t we at least take a stab at it?
If you read a lot of articles in science you know how difficult it is to follow sometimes. There are a lot of acronyms and a lot of words that you don’t really know. There were many times I would read a paragraph, stop to think about it, and have no idea what I just read. Even walking into class I’d have paragraphs highlighted with things I didn’t understand. I became so thankful for the people in my class.
I got to meet an almost entirely new group of people. At a small school like Concordia there isn’t a ton of opportunity to take classes with people who aren’t the same age other than the core credits. Neurochemistry is a capstone class that is taken senior year, considering I am at “senior status” even though I am a junior, I got to take a class with the people I normally just see around the science building.
There were so many different minds and personalities in our small, 14 person class. It was very interesting to hear other people’s perspectives, depending on how they grew up. Every Friday we discussed the issues surrounding our topic that week. For example, what is the cause of Alzheimer’s? Is there a better way to treat it? It seemed that every week it came back to the culture of the busy American’s.
If only we would slow down, take some time to look around. This was something we all had in common. Being very driven, success seeking Cobbers we are pretty much always on the go. FOMO (Fear of Missing Out) is a real thing, fear of missing opportunities, fear of missing fun, and even fear of missing points. A lot of the damage came from oxidative stress. As we are always on the search for the next best thing, the next best cure, maybe it could all be solved by slowing down?
We talked a lot about death and sick people, but not in a sad way. We thought about spending more time with family and friends, making the years count instead of counting the years. As great as finding a cure would be all of us are a few years from being able to be the one who does that, so instead it was mostly about prevention and how to deal with the disease.
So what did I do in my semester of neurochemistry? I got a lot better at reading and communicating science. I figured out how to break down pictures and diagrams into an understandable way. I spent a lot of time listening and understanding other’s opinions. The most valuable lesson I learned from neurochemistry though didn’t have a lot to do with science. This course was rigorous and difficult, but take the time to slow down. The busy life style is a part of our culture and if you don’t keep up you might not make it, but it is important to make time for more than just work. It may save you in the end.
My Capstone Experience
Concordia College implements a capstone course into student’s coursework. It is stated to reinforce the connection between liberal learning and various academic disciplines. Neurochemistry was my capstone course, and I felt it met or exceeded all the exceptions to be called a capstone class.
Instill a love for learning
By taking neurochemistry, it has increased my interest in different areas of study. When I first signed up for the class, I thought it would be another class where the teacher teaches and the student take notes and listen. It was also interesting how it did not have a lab, since every chemistry course I took here had a lab with the lecture.
On day 1, my exceptions were completely thrown out. The class was orientated to helping each other learn. The beginning few weeks had students post on different synapse and signaling molecules. We each had to contribute something to our course wiki page. The fun part was when we got into reading different neurological disease. Mondays had us talk about the paper, and figure what we did not know for “speed dating” on Wednesdays. Friday had us just talk about anything related to the topic of the week. This classroom format was unique and installed a new way for me to learn.
The love of learning also comes from how each of us had to help one another to be able to succeed in this class. The collection of ideas from each other helped pushed thinking into a new aspect, in ways I would never have thought of.
Develop foundational skills and transferable intellectual capacities
Most articles we read each week was a hot topic in health. They included Alzheimer’s disease, Parkinson’s, autism, concussions, and obesity. All of these topics have real world application due to the increase of them in recent years. Concussions have become relevant due to Will Smith’s movie “Concussion” and how retired NFL stars have been seen with chronic traumatic encephalopathy (CTE). All of the topics were known previously before reading the articles, but the science behind how these topics were not uncovered until taking this class.
The knowledge we learned was also transferred to our weekly blog posts on the topic. We were able to condense the article and spread it to the public. Our public service announcement (PSA), also helped spread awareness of a hot topic of our choosing. Both activities helped reinforce our thinking and to be able to tell the public about it.
Develop an understanding of disciplinary, interdisciplinary and intercultural perspectives and their connections
Even though this class was a chemistry class, it was relatable to other disciplinary fields too. Some articles had biological pathways, psychology with animal studies, and English with the blogs. This class presented ideas from different fields in connection with chemistry. This class being neurochemistry was related to neuroscience as well. The study of the central nervous system was always relevant due to these diseases being in the brain.
Cultivate an examined cultural, ethical, physical and spiritual self-understanding
From taking this class, I learned how diverse chemistry can be. With the combination of different disciplinary fields in this course, I learned how they might be able to correlate together to find cures for different diseases. By understanding different pathways from articles in previous weeks, I was able to understand better how they relate to pathways in future articles. It showed me how my area of study could be used in different aspects of the learning.
Encourage responsible participation in the world (BREW)
By taking this class, it helped me think critically in other areas besides chemistry. I also learned how different areas of study can correlate with each other to do good. It made me think more critically about the topics we discussed. Marijuana, which is a schedule 1 drug, has shown to be effective in treating different diseases. The stigma of it still clouds people’s ideas on it.
This class has helped me engage with different aspects of studies, which I would have never known about, or would have thought about.
Autism Spectrum Disorder in Society
The number of children being diagnosed with autism spectrum disorder (ASD) has been consistently increasing the past decade.According to the NIH, It is estimated 1 in 68 children in the United States has autism. This is the highest recorded prevalence in history. Described as a disorder that impairs a child’s ability to communicate and interact with others, autism is a prevalent disorder that modern research needs to focus on to enhance the lives of children everywhere.

There are many common symptoms of autism, but the most distinguishable include impairment of social communication and interactions, or repetitive behavior. A child with autism will fail to respond to their name being called. They might be isolated to their own private world or even engage in repetitive behaviors.
While the causal mechanisms of autism are unknown, modern researchers hypothesis that there may be environmental influences that affect the development of autism in children. Some believe that the inability or culmination oxidative stress in brain may lead to some causes of autism. Implying that the environmental factors. On the other hand, other researchers postulate that some children are predisposed to have fundamental discrepancies with the chemical makeup in their brain.
Overall, there is no concrete understanding or consensus on the disorder. Treatment options also vary with some children receiving chemical agonists or antagonists to excite or inhibit various neurological functions. On the other hand, others seek therapy options to learn how to handle the condition and work towards full immersion into society.
Personally, I find the ASD to be a fascinating topic. I think it encompasses all of the neuronal pathways we covered in this class and proved to be a difficult, yet satisfying problem to try and solve. I hope that the field of neuroscience will continue to unveil the ASD story so we can learn, as a society, how to treat, diagnose, and prevent this disorder.
Autism Spectrum Disorder
Autism is described as a disorder that impairs a child’s ability to communicate and interact with others. The number of children being diagnosed with autism spectrum disorder (ASD) is slowly rising. According to the NIH, It is estimated 1 in 68 children in the United States has autism. The exact number is unknown, but it is still a serious problem that needs to be handled.
Symptoms of autism include impairment of social communication and interactions, or repetitive behavior. A child with autism will fail to respond to their name being called. They might be isolated to their own private world or even engage in repetitive behaviors.
The CDC, NIH and Mayo Clinic state the causes of autism may be due to genetic factors and environmental factors. Some genes may make a child more susceptible to autism. There are over 100 genes that may induce some symptom the autism spectrum. Currently, there is no identified environmental cause related to autism, but environmental factors may cause abnormal gene function and development.
What truly causes autism in the brain is more complicated than genetic or environmental factors. The signaling pathway associated with ASD is complex. From this figure, it shows the possible pathway ways ASD may be triggered. The figure may show which molecules are mutated in red, but the problem lies with what molecules started ASD first? With the mRNA portion in the middle of the figure, PTEN, TSC1 or Shank each can cause the increase of activation of FMRP and eLF4E. This will result in too much gene translation, which could lead to ASD.
Neuroligins (NLGN) are synaptic cell adhesion molecules at the post-synaptic membranes. They help mediate synaptic functioning and maintenance of synapses between neurons. Dysfunction of NLGN impairs the properties of synapses and disrupt neural networks. This leads to an imbalance of excitatory and inhibitory signaling, which could lead to ASD.
There are medications available in treating symptoms of autism, but no cure exists. As research continues on ASD, the pathways are a potential in trying to understand ASD, then a cure may be found.
I am glad I was wrong…
As an international student, I had no idea what a capstone course means. When I first heard the word “capstone”, I thought it was just a word that I did not know. And well, I knew nothing about a capstone course. At first, when I registered, I expected that the Neurochemistry class would be just hard, with a lot of memorization and reading.
I am glad that I was wrong.
Even before the semester begin, the professor invited the students to explain what we should expect of the capstone course. I got really excited, because it would be a chance to not just learn about Neuroscience, but to TALK and DISCUSS about it.
Reading through Concordia goals for liberal learning, I just thought that Neuroscience is the perfect scientific area to put all of them into practice.
- Instill a love for learning
- Develop foundational skills and transferable intellectual capacities
- Develop an understanding of disciplinary, interdisciplinary and intercultural perspectives and their connections
- Cultivate an examined cultural, ethical, physical and spiritual self-understanding
- Encourage responsible participation in the world
I mean, who would not love learn more about what is going on in the brain and how it works? (Ok, maybe some people would not, but I know I love it)
With the schema that the professor created, where every week all the students had to present more information about some small part of a topic, I believe that we all improved our skills in transferring intellectual capacities. We were not only students, we ended up being teachers, sharing what we learned with the whole class.
Neuroscience, a scientific area that involves biology, psychology, data processing, chemistry, nutrition, and so on. Learning about the brain instigates you to make the connections between all these subjects. It is an interdisciplinary course, and I dare say that is the most interdisciplinary of all.
I believe that we have to unite all the disciplines in order to better understand the complex structure that the brain is.
Understanding and learning about the brain is to understand and learn about ourselves. It is to unveil our behaviors and beliefs, seeking out a way to improve our society.
Responsible participation in the world is what neuroscience is about. In our class, we discussed every week how brain diseases affect our society and how we could treat them. Alzheimer’s disease, Parkinson’s disease, Schizophrenia, Bulimia, Obesity, Autism, Alcohol and Nicotine addiction. All these diseases and even more were discussed in each week, what made me realize how much neuroscientists are working in order to understand how we could help people that are suffering with brain diseases.
I am glad that I took this capstone course as a part of my year in Concordia. I would never have the chance to do such a different and exciting class back in Brazil. In the end, it was not just a class, it was an opportunity to learn and appreciate science, improving my communication and research skills. I will leave Concordia proud and happy to have participated in this great experience that was Neurochemistry class.
Sarcosine: Good News for Schizophrenia
What is Schizophrenia?
Schizophrenia is a developmental brain disorder in which people interpret reality differently than normal. This can result in hallucinations, delusions and disordered thinking and behavior.
What causes Schizophrenia?
A recent review article cites the Glutamate signaling system within the brain. This is the main excitatory pathway in the brain and it is believed to be overactive in the brain of people with schizophrenia. This over excitation causes normal stimuli to often be too much for people with schizophrenia to handle.
What is Sarcosine and what does it do?
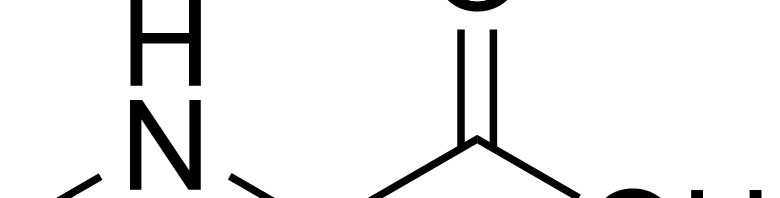
Sarcosine is actually an amino acid that is naturally found within the human body. But besides just being an amino acid it also blocks glycine transporters in the brain. You see one of the main receptors in the Glutamate signaling system is the NMDA receptor. This receptor needs not only glutamate to fire but also needs to be in the presence of glycine. Without a transporter to bring the glycine to the NMDA receptor it cannot do its job. However in the case of schizophrenia the NMDA receptors appear to be working overtime and blocking the glycine transporter helps the receptors get on a regular schedule. On paper one would think that sarcosine would help the symptoms of schizophrenia and studies also agree. The cool thing about sarcosine is that it treats negative symptoms. Schizophrenia symptoms are broken up in to positive and negative symptoms. The positive symptoms are the hallucination type and the negative symptoms are essentially the cognitive decline part of schizophrenia. Until recently most treatments only treated the positive symptoms but studies show that taking sarcosine along with normal treatment can improve negative symptoms as well.
Parents: When is it Appropriate for Your Kids to Drink?
There is no doubt about it – alcohol is a big part of our society. Whether someone drinks or not is actually, partly a defining characteristic about them. Maybe that’s purely form the perspective of a college student, but I think there’s some merit in saying that. And being in college, I have definitely seen the effects of drinking, whether they are positive or negative. Obviously, drinking is mostly a harmless event that is usually enjoyed with a group of friends, but there are significant dangers as well. Through my experiences with drinking, however, there is a commonly asked question: when did you start drinking? In high school? Freshman year? Never?

Usually these groups are split. Some people started drinking and partying in high school with their high school friends, usually behind the backs of their parents. Some didn’t drink until college. And some have never had a drink before. Whatever group a person belongs in, I’ve noticed a trend – those that drank in high school have tended to binge drink at a dangerous rate less often in college. Seems crazy and obviously secluded to the small sample size that is Concordia, but it’s my observation nonetheless. So, should that be encouraged? Have a drink or two with your parents before coming to college? Or even party a couple times with your high school friends before you get to college? It’s an interesting concept, that’s for sure.
Why exactly would it make sense to “condition” someone to drinking before they get to college where it is so prevalent? Well, as I mentioned before, binge drinking is common in college. And obviously harmful. However, if someone is used to drinking or has done it before, binge drinking becomes less prevalent. That would mean the effects of binge drinking on the brain would also decrease if an incoming freshman had experienced drinking before.
What exactly are those effects? Well, it’s well defined that alcohol has a direct impact on certain parts of the brain that involve movement, speech, judgment, and memory. The impact causes impairment, which results in the typical drunken behavior such as slurred speech, difficulty walking, blacking out, and impulsive behavior. In the case of a binge drinking session, these effects are amplified significantly.
The question becomes, should parents drink with their kids before they go off to college? In my experience, it seems that there would be advantages to this thought process. However, it is completely dependent on the parents.
A Reflection on the Journey: Neurochemistry Capstone
By far this course has been one of my favorite at Concordia. I looked forward to going to Neurochemistry every day.
This course was a place where we could put our chemistry expertise to use and explore real world problems. It was so fun to have discussions surrounding real world science with fellow students and be able to apply our various backgrounds to the discussion.
Throughout the course we delved into various neural disorders that worked to incorporate the five goals of liberal learning Concordia has set forth as the cornerstone for capstone experiences.
Instilling a Love for Learning
Each week we explored a different neurological disease. Each week we worked to understand the science of the disease with each other through discussion Mondays and speed dating Wednesdays.
The love for learning was so easily instilled as we worked together to understand each piece of the puzzle. Not only was it immediately interesting to us all, but it was collaborative learning that made the class so exciting.

The atmosphere was full of students who cared about the subject material and were excited about learning all we could about the disease and the best ways to treat and/or cure it.
The exciting real life learning we did in neurochemistry instilled in us a passion for learning and discussing the things we learned with one another, and also to share what we had learned with the larger population through our blogging.
Developing Skills and Transferable Intellectual Capacities
What made this course so great was the ways in which it was applicable to daily life. We all had heard of the diseases we discussed, and had an interest in figuring out what chemically was causing the diseases.
By having these vested interests, it was easy to study the diseases in depth and then use the knowledge we acquired to write blogs to share the information with the broader community.
Blogging was one of my favorite parts of this course. It was a chance to put the complexities of chemistry we had researched throughout the week into an understandable story for people outside of the Cobber Chemistry Bubble to engage.

The skills we developed will be so useful in our professional careers as we are required to explain complex science to people who have a various array of scientific literacy.
As a physician in particular, I find the skills I have developed in this course to be essential to relating to patients and making them feel comfortable in their health journeys.
Developing Diversity of Disciplines
Our class had a wide array of expertise. Many of us were chemistry majors, but we also had several biology majors and psychology majors. This variety aided in our collaborative learning immensely.
I have very little understanding of psychology, and very little interest in it. By those who live and breathe psychology were so helpful to me.
They were the ones who understood the papers about schizophrenia, bulimia nervosa, and autism best. They were the ones who could explain where clinical treatments currently lie, and speak from experience in working with individuals who suffer from these diseases.
In this way, the course has instilled in me the importance of interdisciplinary work, and the invaluable resource that is new perspectives.
It is always humbling to be around someone who’s strengths differ from your own. I was blown away by the different perspectives in this class and how they shed light to the problems we were discussing. They helped me to understand the diseases and solutions in new and deeper ways.
The interdisciplinary work in this course helped me to understand my own abilities and where they are helpful as well as where they fall short in looking at health concerns. I will carry this awareness and value with me as I venture into the world of medicine.
Cultivating Holistic Self-understanding
As I have already touched on, the interdisciplinary work we did in this course helped me to understand how my training in chemistry has both prepared me, and in some ways hindered me in my abilities to solve problems.
With a chemistry brain I am very good at deduction and problem solving, but terrible at memorizing specific bits of information for each disease. In this way, I relied heavily on my biology and psychology classmates to fill in the missing pieces.
It was also through this course and collaborative learning that I became aware of my great training in taking lots of information and organizing it into a helpful and useful format.
Inspiring BREW

As an aspiring physician I found this class to be so applicable to my future career in medicine. I found the ways of discussing both the science and the societal norms surrounding each disease we looked into to be asking the deep questions of what this information means to the world.
From this experience I have gained the ability to critically engage scientific research and the ways in which understanding the deep chemistry of the problems can help in developing treatments and prescribing medicines.
As a chemistry major, graduating from Concordia College in just four short days, I feel so prepared to be a thoughtful and informed responsibly engaged participant in the medical profession, both locally and globally. Both this course and all those I have taken at Concordia have prepared me so well for the future ahead.