You dive for the ball, just barely have it in your hands, when you collide head-first with the player from the other team! You pick yourself up off the ground, shake off the blow, and keep going. You’re wearing a helmet, so you’re fine, right? Beyond your helmet, under your skull, zoom in on your brain, and now you have tiny pores in your lipid membranes. A rush of sodium and calcium enter your brain cells. The neurometabolic cascade has begun.
The Science: The New Neurometabolic Cascade of Concussion
Traumatic brain injuries, like a head-first collision during football, cause abnormal ion flux, the moving of ions across the cell membrane. Specifically, large amounts of calcium go into the cell, leading to phosphorylation and structural issues in the axons and cytoskeleton of neurons. The large amounts of calcium, as well as sodium entering the cell, cause rapid depolarization, leading to repeated glutamate release, the main excitatory neurotransmitter. This cascade of signaling disrupts the ionic balance of the cells, and in an attempt for the ionic pumps to restore homeostasis, they use too much ATP. This puts your brain cells into an energy crisis, which is further exacerbated by the excess calcium being sequestered to the cell’s mitochondria to try and help the ionic balance. Now the mitochondria are dysfunctional, and that definitely doesn’t help the energy crisis. This entire cascade of events leads to the activation of your brain’s microglia, cells of your immune system that respond to the injury. This leads to brain inflammation [1].
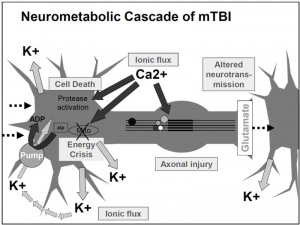
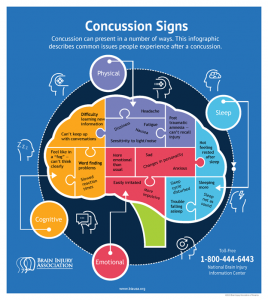
So the ions in your brain are currently out of balance, causing you to have a migraine, along with sensitivity to light and sound. The injuries to your axons are leaving you with impaired cognition, slowed processing speed, and slow reaction time. So much for that game-winning maneuver you were planning. You are in an energy crisis, which is leaving you vulnerable to a second injury, and you’re right back out on the field.
Long-Term Impacts: Chronic Traumatic Encephalopathy
Let’s say you get through this game with minimal effects from your earlier collision, but in the next game, when you score the winning touchdown, you crash into the end zone head-first. Then a couple of games later, you get tacked to the ground, and your head feels like it’s vibrating from how hard it smashed against your helmet. This time, when your mitochondria are zapped of energy, your axons are injured, and your microglia are mad at you for making them work overtime, the neurometabolic cascade leads to more harrowing long-term impacts. The metabolic changes last longer, and trigger intracellular proteases, which can cause the cascade to lead to apoptotic cell death [1].
Chronic Traumatic Encephalopathy is a neurodegenerative disorder caused by repeated injury to the brain. Symptoms include cognitive impairment, behavioral changes, mood disorders, and motor symptoms [2]. CTE has been linked to contact sports such as football [3], and studies are showing that more than one-third of football players believe that they are impacted [4].
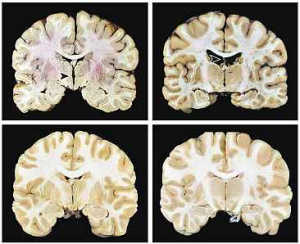
These acute and chronic dangers of traumatic brain injuries are even more frightening in kids before their brains have finished developing. When the brain is injured during critical periods of development, it can impact the brain’s ability to function normally [6]. This means it’s even more crucial for return to play and other guidelines to take into consideration the science behind repeated head injury. Or even better, the guidelines around protecting our brains during contact sports, biking, riding in vehicles, and other activities need to be stricter. Should kids with delicate, developing brains really be playing brutal contact sports like football, soccer, or boxing? Why can’t society modify these activities to prevent head collisions, and make safer helmets? We need to continue research into the long-term impacts of traumatic brain injuries and use that science to make informed decisions regarding contact sports, especially in developing brains.
References
[1] Giza, C. C., & Hovda, D. A. (2014). The new neurometabolic cascade of concussion. Neurosurgery, 75(Supplement 4), 24–33. https://doi.org/10.1227/neu.0000000000000505
[2] Mayo Foundation for Medical Education and Research. (2023, November 18). Chronic traumatic encephalopathy. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/chronic-traumatic-encephalopathy/symptoms-causes/syc-20370921
[3] U.S. Department of Health and Human Services. (2023, August 28). CTE identified in brain donations from young amateur athletes. National Institutes of Health. https://www.nih.gov/news-events/news-releases/cte-identified-brain-donations-young-amateur-athletes
[4] Study of Former NFL Players Finds 1 in 3 Believe They Have CTE. Mass General Brigham. (2024, September 23). https://www.massgeneralbrigham.org/en/about/newsroom/press-releases/study-finds-1-in-3-former-nfl-players-believe-they-have-cte#:~:text=A%20new%20study%20of%20nearly,mortem%20exam%20of%20the%20brain.
[5] U.S. Department of Health and Human Services. (2023b, September 19). Chronic traumatic encephalopathy in young athletes. National Institutes of Health. https://www.nih.gov/news-events/nih-research-matters/chronic-traumatic-encephalopathy-young-athletes
[6] Blackwell, L. S., & Grell, R. (2023a). Pediatric traumatic brain injury: Impact on the developing brain. Pediatric Neurology, 148, 215–222. https://doi.org/10.1016/j.pediatrneurol.2023.06.019


 [3]
[3]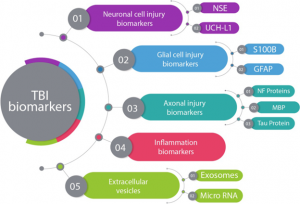 [5]
[5]
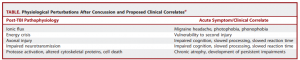
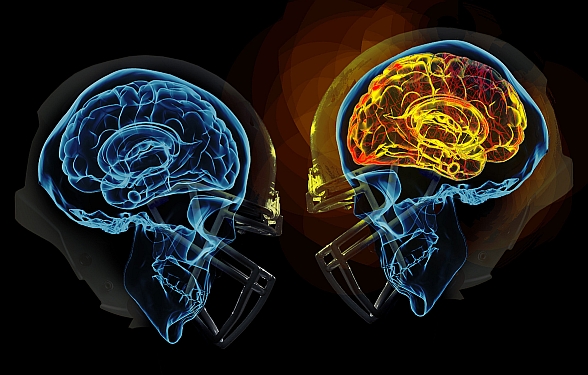


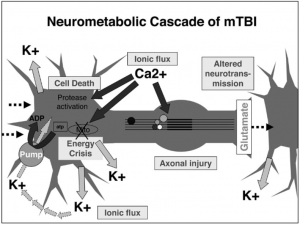

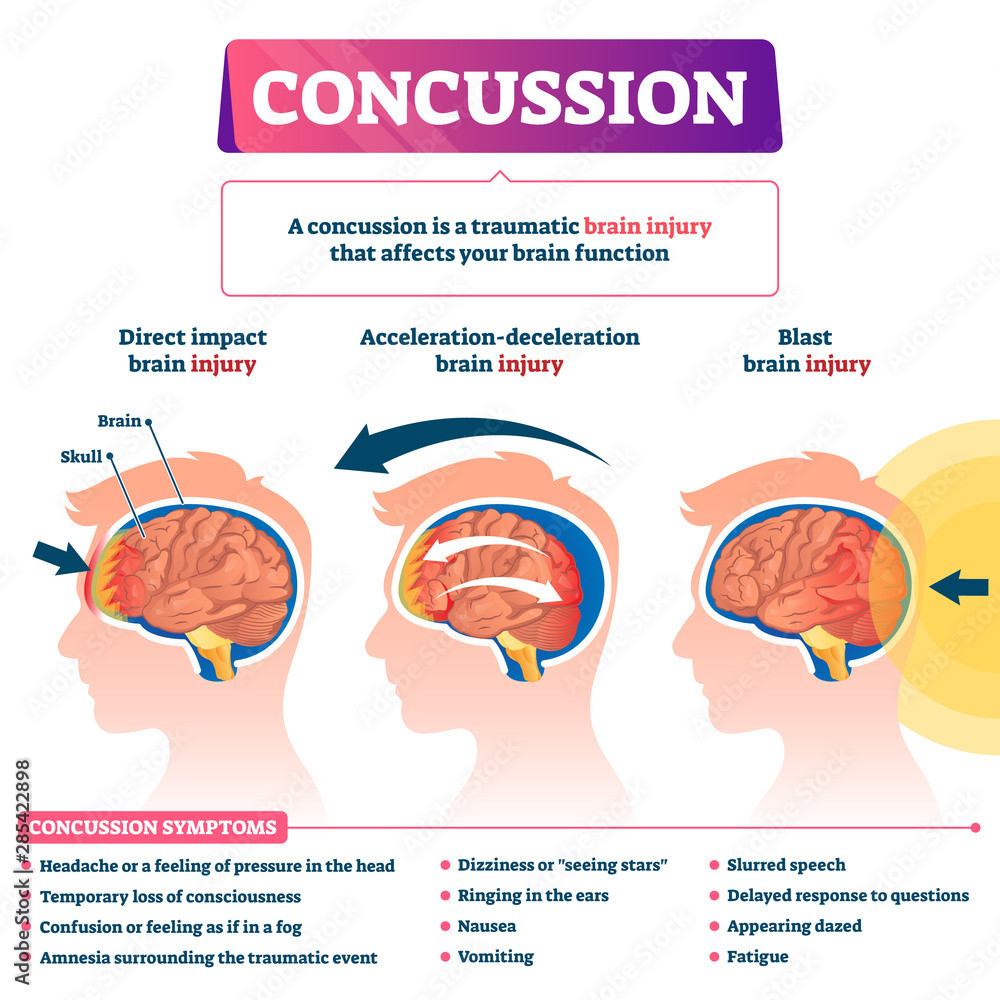



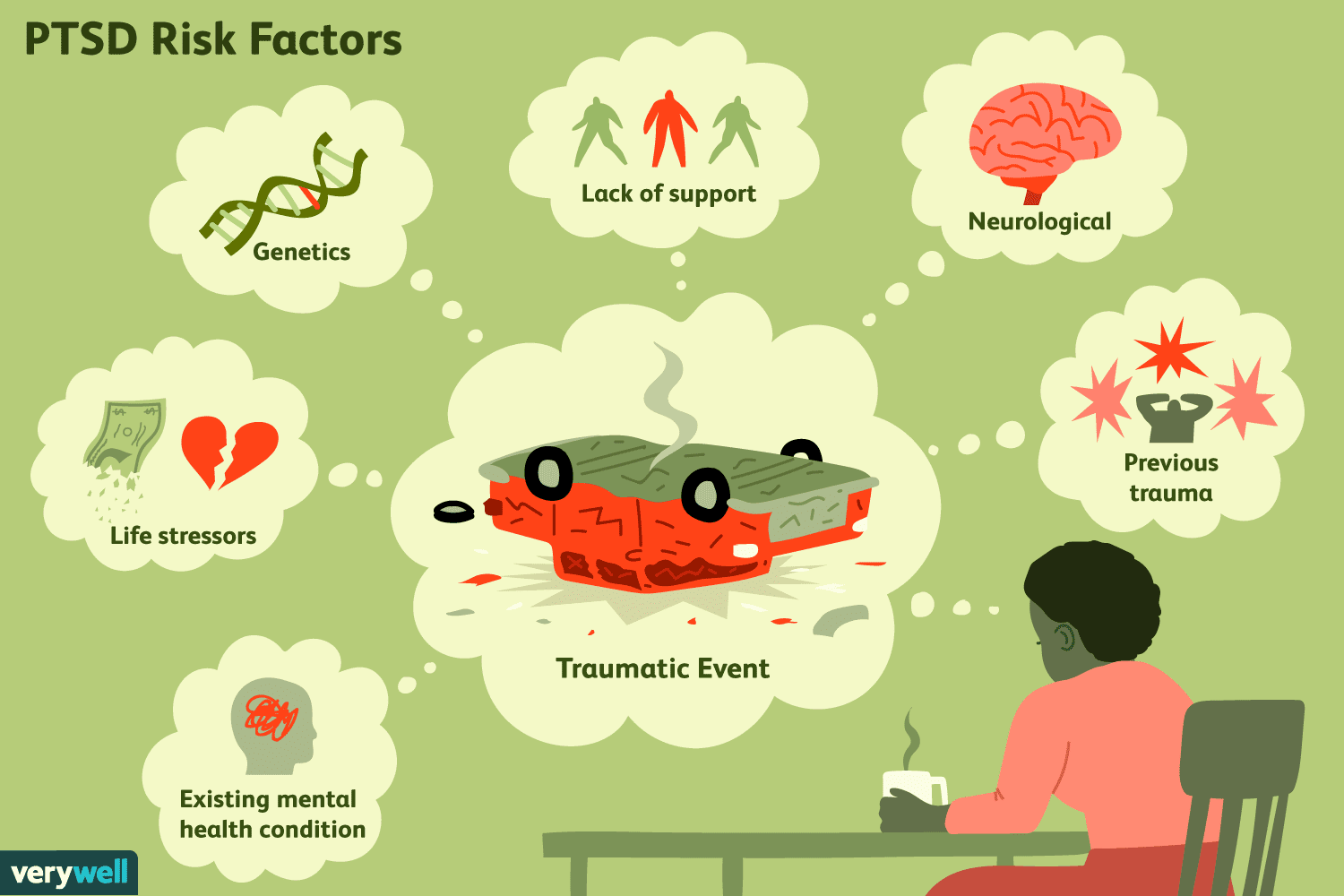 Figure 1. Diagram showing past events that could potentially lead to cases of anxiety disorders such as Post Traumatic Stress Disorder (PTSD) [2]
Figure 1. Diagram showing past events that could potentially lead to cases of anxiety disorders such as Post Traumatic Stress Disorder (PTSD) [2]