From Wnt to Wonder → The Neuroscience Behind Schizophrenia
Schizophrenia is a complex and misunderstood disorder that affects emotional and physical behavior. Even though this disorder affects millions, it remains a poorly understood developmental disorder. The stigma that co-occurs with Schizophrenia leads to oversimplified symptomology.
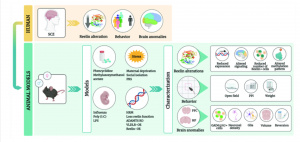
However, emerging research reveals Schizophrenia is strongly linked to the Wnt pathway, which is a network of proteins that work together in brain developement, communications, and homeostasis. Research suggests that dysregulation of the Wnt pathway could be the root of Schizophrenia, leading to the importance of understanding what Schizophrenia is and how it manifests.
What is Schizophrenia?
This disorder is multifaceted; affecting thought processes, emotions, and behaviors. There are many misunderstandings and stigmas surrounding Schizophrenia. Characteristics in symptomology lead to discrimmination which overshadows the biological and developmental beginnings of the disorder.
There are five different subtypes of Schizophrenia. The symptoms for each subtype vary, and understanding these subtypes could be helpful in recognizing the symptomology and how it connects to the biology beneath the surface.
- Paranoid: Most common form: consists of hallucinations and/or delusions, speech and emotions may not be affected, develops later in life.

- Disorganized/Hebephrenic: Develops 15-25 years, disorganized behavior, and thoughts, short lasting hallucinations/delusions, may have diagnosed speech patterns and others may find it difficult to understand you.
- Catatonic: Move from being very active to very still, may not talk much, can mimic others speech and movement.
4. Undifferentiated– some signs of each
5. Residual -history of psychosis, only negative symptoms, slow movement, poor memory, lack of concentration, and poor hygiene
What is the Wnt Pathway?
One pathway has emerged as a critical path in Schizophrenia: the Wnt pathway. This is a network of proteins that communicate signals through cellular processes and facilitate regulation and communication throughout the brain. Methods have uncovered that dysregulation of the Wnt pathway is linked to multiple developmental disorders, contributing to cognitive deficits, structural brain changes, and symptomology. Structural brain changes cause alterations in neural connectivity that contribute to cognitive deficits and emotional dysregulation.
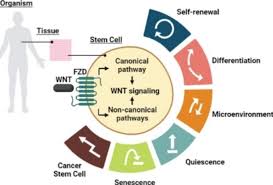
The Wnt pathway is used in a range of biological processes such as cell development, homeostasis, and differentiation which is why the pathway is so critical in psychiatric disorders. All processes stimulate each other, the Wnt pathway can be evalutated to understand why symptoms occur in Schizophrenia and ways to prevent them.
Think of the Wnt pathway as a series of relay runners passing a baton (signals) to regulate brain processes. When something disrupts the relay, its like dropping the baton- leading to the changes, neurological and behavioral, that we see associated with Schizophrenia.
The Science of the Wnt Pathway
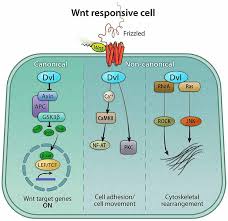
The Wnt pathway is essentially a network of Wnt proteins, which are a family of secreted glycoproteins that bind to receptors on the surface of cells which in turn initiates a cascade of signaling events. Frizzled (Fz) receptors are the primary receptors for Wnt proteins. When Wnt binds to Fz receptors, intracellular signaling cascades are activated and trigger various cellular responses. Two pathways that can be trigged are a canonical pathway and a noncanonical pathway. Beta catenin is involved when a canonical pathway is trigged by an intracellular signaling cascade. On the other hand, when a noncanonical pathway is triggered, beta catenin is not involved in the process.
How it works: Beta Catenin
β-Catenin is a central component of the canonical Wnt signaling pathway, which regulates a wide variety of cellular processes such as cell differentiation, proliferation, migration, and synaptic plasticity. It acts as a transcriptional co-activator that, in the presence of Wnt signaling, translocates to the nucleus and binds to T-cell factor/lymphoid enhancer factor (TCF/LEF) transcription factors to initiate gene expression.
Altered β-catenin signaling during critical periods of brain development, especially in the formation and maintenance of neural circuits, may contribute to cognitive deficits and impaired synaptic plasticity seen in Schizophrenia. β-Catenin’s role in regulating synaptic function might be disrupted in individuals with Schizophrenia, affecting neuronal connectivity and circuit formation.
Two Important Pathways
The Wnt pathway is critical for optimal brain health. By understanding the relationship between the Wnt pathway and Schizophrenia we can see a potential future of treatment possibilities. Research proposes that interventions of the Wnt pathway could improve life quality for those with Schizophrenia.
Canonical
The canonical pathway produces beta catenin. This pathway is initiated when Wnt ligands bind to receptors on the cell membrane, primarily a Fz family receptor with low-density lipoprotein receptor-related proteins. The pairing of these two molecules sets off a chain reaction inside the cell.
Without Wnt signaling (when it’s turned off), beta catenin is broken down by the destruction complex, which is a group of proteins involving, GSK3β, Axin, APC, and CK1α. This group of proteins stays assembled when Wnt signaling is off. If Wnt ligands are binding to their receptors (Wnt signaling is on) the complex is disassembled. Therefore, beta catenin is accumulated in the cytoplasm and moves to the cell nucleus. Beta catenin then has the ability to interact with co-factors like TCF/LEF to initiate the transcription of Wnt-dependent target genes.
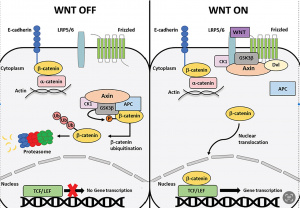
Fig. 4 Comparison of Wnt signaling off/on
Noncanonical
- Does not involve β-catenin-mediated transcription
- Two pathways are the planar cell polarity (PCP) and Wnt/calcium pathways
- PCP signaling involves Wnt signaling through Fz receptors and G proteins
- Rho and Rac proteins are activated to regulate the cytoskeleton
- PCP signaling involves Wnt signaling through Fz receptors and G proteins
- The Wnt/calcium pathway is triggered by Fz activation
- Increases intracellular Ca2+ levels
- Leads to the activation of protein kinase C (PKC), affecting a broad range of cellular functions
- Particularly in the central nervous system (CNS) which influences neural circuit formation and synaptic plasticity
- Leads to the activation of protein kinase C (PKC), affecting a broad range of cellular functions
- Increases intracellular Ca2+ levels
What’s the big whoop?
Schizophrenia is a complex psychiatric disorder with significant neurodevelopmental and genetic groundwork. By understanding the inner-workings of Wnt signaling, research can open new innovative therapies that potentially could change the lives of individuals with Schizophrenia.
Recent research has highlighted the critical role of the Wnt signaling pathway, particularly β-catenin, in the development and progression of Schizophrenia. β-Catenin is a key player in the canonical Wnt pathway.
β-Catenin and the Wnt signaling pathway play a significant role in brain development, synaptic function, and cognitive processes. Dysregulation of this pathway, particularly through altered β-catenin signaling, is emerging as an important factor in the development of Schizophrenia. As research continues, understanding the precise role of β-catenin could open new doors for targeted therapies, offering hope for better treatment strategies for this complex and debilitating disorder.
By exploring the intersection of genetic, molecular, and pharmacological factors in Wnt signaling, we move closer to understanding the biological basis of Schizophrenia and developing more effective treatments.
References
KK;, S. (n.d.). An emerging role for Wnt and GSK3 signaling pathways in Schizophrenia. Clinical genetics. https://pubmed.ncbi.nlm.nih.gov/23379509/
Liu, J., Xiao, Q., Xiao, J., Niu, C., Li, Y., Zhang, X., Zhou, Z., Shu, G., & Yin, G. (2022, January 3). Wnt/β-catenin signalling: Function, biological mechanisms, and therapeutic opportunities. Nature News. https://www.nature.com/articles/s41392-021-00762-6
MacDonald, B. T., Tamai, K., & He, X. (2009). Wnt/beta-catenin signaling: components, mechanisms, and diseases. Developmental cell, 17(1), 9–26. https://doi.org/10.1016/j.devcel.2009.06.016