In a quiet neuroscience lab lit by the blue glow of bioinformatics screens, a group of researchers stared at a common enemy—one that doesn’t knock, doesn’t wait, and doesn’t often lose. Glioblastoma. A tumor so aggressive and elusive that even with today’s best medicine, it often wins. But this time, the researchers weren’t trying to kill the tumor directly. They were trying to listen to it.
Because cancer, like everything in our body, talks.
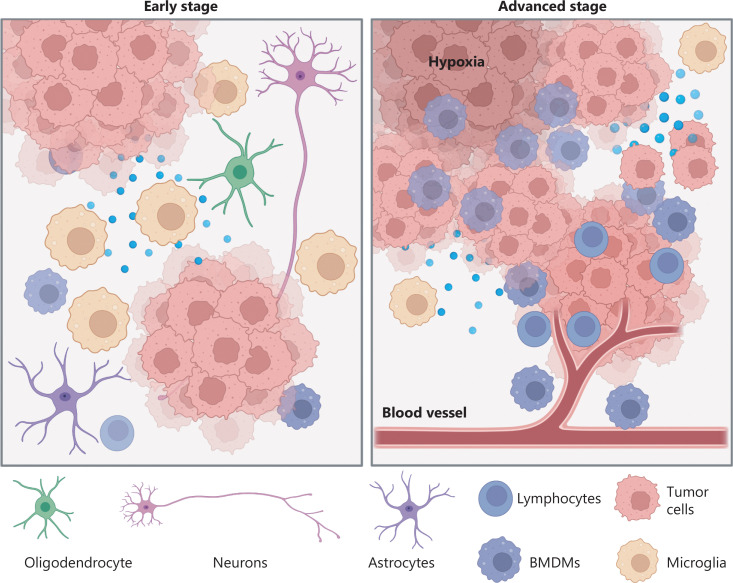
 Abstract by Alisha Debleye depicting the tumorous and dangerous growth in the human brain that can sometimes go untreated or undiagnosed.
Abstract by Alisha Debleye depicting the tumorous and dangerous growth in the human brain that can sometimes go untreated or undiagnosed.
It communicates using intricate molecular “languages” called signaling pathways—chemical networks that tell our cells when to grow, when to rest, and when to die. For healthy cells, this communication keeps everything running smoothly. But glioblastoma doesn’t play by the rules. Instead, it hijacks these pathways, manipulating messages to fuel chaos instead of harmony.
That’s what the article “Understanding and Exploiting Cell Signalling Convergence Nodes and Pathway Cross-Talk in Malignant Brain Cancer” is all about. It’s not just about what makes GBM grow. It’s about how GBM outsmarts nearly every treatment we throw at it—by rerouting, rewiring, and repurposing the very systems that keep our cells alive.
The Whispering Networks of GBM
Our bodies use three key signaling pathways to manage cell behavior: MAPK, PI3K, and cAMP. Think of them as information highways: MAPK fuels growth, PI3K ensures survival and movement, and cAMP acts as a cellular checks-and-balances system.
Figure 1 shows the MAPK pathway being activated by receptor tyrosine kinases, sending messages for tumor growth, survival, and movement. This pathway is often hyperactive in GBM, particularly when NF1—a negative regulator—is lost.
In glioblastoma, these systems are corrupted. The tumor amplifies MAPK and PI3K to accelerate growth and invasion, while it silences cAMP—the very pathway that normally tells damaged cells to self-destruct.
The PI3K pathway is slightly different. This is how it works.
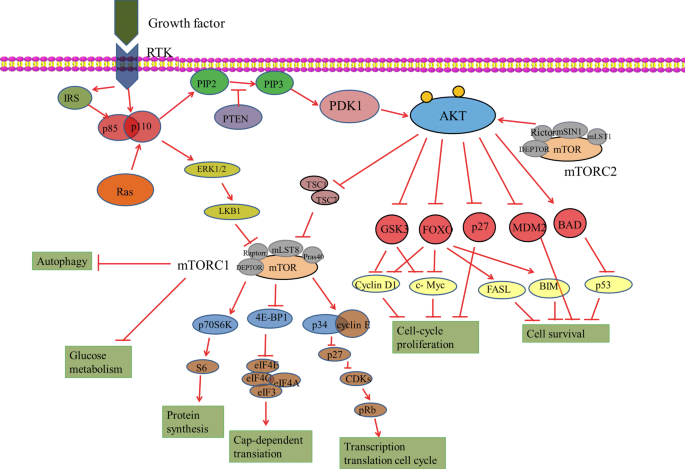
Figure 2 illustrates the PI3K pathway, another powerhouse of tumor survival. It activates downstream molecules like Akt and mTOR, which promote cell proliferation and metabolic rewiring. Together, PI3K and MAPK form a dangerous duo.
It even discusses how the GPCR pathways, specifically cAMP pathway is effected.
Figure 3 depicts the cAMP pathway—a tumor suppressor system that is unusually suppressed in GBM. Normally, this pathway promotes cell death and inhibits uncontrolled growth. But glioblastoma cells lower cAMP activity by reducing the enzyme adenylyl cyclase or increasing phosphodiesterases (PDEs) that degrade cAMP.
But these pathways don’t work in isolation.
Figure 4 shows how all three signaling networks converge on CREB, a central transcription factor. CREB integrates signals and activates genes responsible for growth, invasion, and immune evasion. It’s like the tumor’s master conductor—one that researchers now see as a promising therapeutic target.
But Why Should We Care About This At All?
Duh, Because its cancer! Also, GBM is the deadliest brain cancer. Its median survival rate is around 14 months, and fewer than 5% of patients survive five years with the disease1. It doesn’t just grow—it infiltrates, migrates, and recurs. It touches memory, movement, and personality. It devastates not only the patient but everyone who loves them.
But the science we read this week gives us hope. The review showed that targeting convergence nodes—places like CREB where multiple pathways meet—might finally outmaneuver the tumor. Drugs like BKM120, a pan-PI3K inhibitor, and Vemurafenib, which targets RAF in the MAPK cascade, are being tested in clinical trials2. Meanwhile, drugs like forskolin and PDE inhibitors, which restore cAMP signaling, show promise in triggering apoptosis, or programmed cell death3.
In fact, when researchers combined a cAMP activator with a MAPK inhibitor, some previously resistant GBM cells finally died4. This is a huge breakthrough. It suggests that combination therapy, hitting multiple pathways or convergence points at once, could finally disrupt the tumor’s carefully stolen communication.
What Can Society Do to Help The Cause?
Awareness is the first weapon. Glioblastoma symptoms often appear suddenly and worsen quickly. If you or someone you love experiences persistent headaches, personality changes, nausea, vision problems, or seizures, it’s important to seek medical attention quickly5. While there’s currently no routine screening for GBM, early diagnosis through MRI can sometimes buy precious time for surgery and treatment6.
Advocate for funding. Research into GBM receives a small fraction of overall cancer research funding7. By supporting organizations like National Brain Tumor Society or American Brain Tumor Association, the public can help fuel new discoveries—like the ones we studied this week.
And most importantly, don’t look away. Brain cancer may be rare, but its impact is massive. The more we learn about how it communicates, the better we can prepare, detect, and defeat it. We are on the cusp of a revolution in brain cancer treatment—not because we’ve found a cure, but because we’ve learned how to listen
Footnotes: