Artstact created by K. Leppke with help of ChatGPT
Neural Stem Cells and Development
The development of your brain was an extremely complex process, which was critical to the way your brain is structured and functions today. At the center of this process early on are neural stem cells, NSCs. NSCs helps neurons develop, get organized, and form synapses. They follow a very specific set of biological rules to create the brain’s complicated structure. If development strays from these rules, the alterations to the brain can lead to consequences can last a lifetime.
But unfortunately, NSCs are influenced by both genetic factors and environmental impacts. Small errors in this early stage of brain development can cause a ripple, effecting more major changes downstream. Early environmental factors, like prenatal infection, stress, inflammation, or poor nutrition, can effect NSC processes. If specific genes don’t function properly, their instructions for the brain can cause poor neuron structure. The RELN and DISC1 genes typically help guide how stem cells grow, divide, connect, but their deletion or nonfunction has been linked with schizophrenia (1).
The Schizophrenic Brain
Signaling pathways in the brain, like Wnt and GSK3, play a crucial role in regulating NSC survival and function. These pathways help neurons form correctly and establish strong synaptic connections, supporting cognition and behavior. Abnormalities in Wnt signaling and GSK3 activity can interfere with NSC survival and differentiation (2). This can lead to fewer neurons or non-functioning neurons, with weaker or less organized connections. These changes in Wnt and GSK3 activity ultimately affect essential processes for major brain functions, like thinking, memory, and perception.
Several genetic factors associated with Schizophrenia can alter the behavior of NSCs and disrupt the formation of neural circuits. Therefore, researchers have been experimenting with how these genes impact early development with NSCs. Genes such as RELN and DISC1, and others involved in Wnt and GSK3, help regulate NSC processes. When these genes are altered or mutated, NSCs may divide improperly or fail to generate functional neurons, leading to reduced synapse formation and weaker connectivity (1). An experiment was done to see how the RELN gene impacts stem cells becoming functional neurons. This experiment, and this figure, identifies RELN as a key contributor to poor neuronal connections (3).
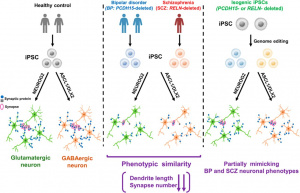
Figure 1: This diagram shows how stem cells were reprogramed into induced pluripotent stem cells (iPSCs), which act like early NSCs and can develop into many types of brain cells. These iPSCs are then directed to become glutamatergic neurons and GABAergic neurons. Neurons derived from schizophrenia patients, with a RELN deletion, showed neuron abnormalities, indicating poor neuronal connectivity. To confirm that these changes are genetic, researchers deleted the same genes in healthy stem cells, which partially reproduced the same neuronal defects. This shows how genetic factors effect neural development and synaptic structure in schizophrenia (3).
Conclusion
Disruptions in early developmental pathways, specifically Wnt and GSK3, have lifetime effects on brain structure and function, which can contribute to the development of Schizophrenia. These pathways regulate NSC growth, differentiation, and connectivity, meaning that slight alterations during early brain development can lead to poor synaptic networks and neural communication. Therefore, understanding and potentially targeting these pathways offers a promising direction for earlier intervention and more effective treatments that address the underlying biological causes.