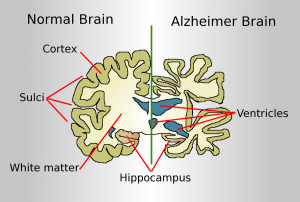
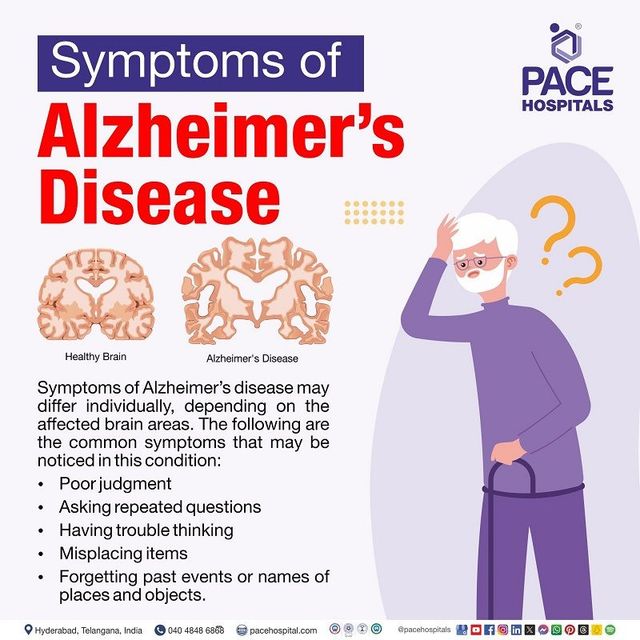
Alzheimer’s disease and Polycystic Ovarian Syndrome (PCOS) are two seemingly unrelated conditions that disproportionately affect women. Alzheimer’s, a devastating neurodegenerative disease causing symptoms such as memory loss and cognitive decline, is the leading cause of dementia and affects over twice as many women as men[1]. PCOS, on the other hand, is a common hormonal disorder that impacts 1 in 10 women of reproductive age, causing symptoms like irregular periods, weight gain, and fertility issues[2]. While these conditions appear distinct, emerging research suggests they may be linked by a shared underlying mechanism: metabolic dysfunction.
What is “Type III Diabetes”?
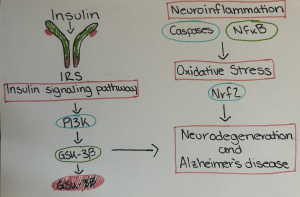
The term “Type III Diabetes” describes the link between insulin resistance in the brain and Alzheimer’s disease. The 2020 review “Insulin signaling pathway and related molecules: Role in neurodegeneration and Alzheimer’s Disease” highlights how impaired insulin signaling in the brain contributes to the accumulation of amyloid plaques and tau tangles, leading to Alzheimer’s disease progression[3].
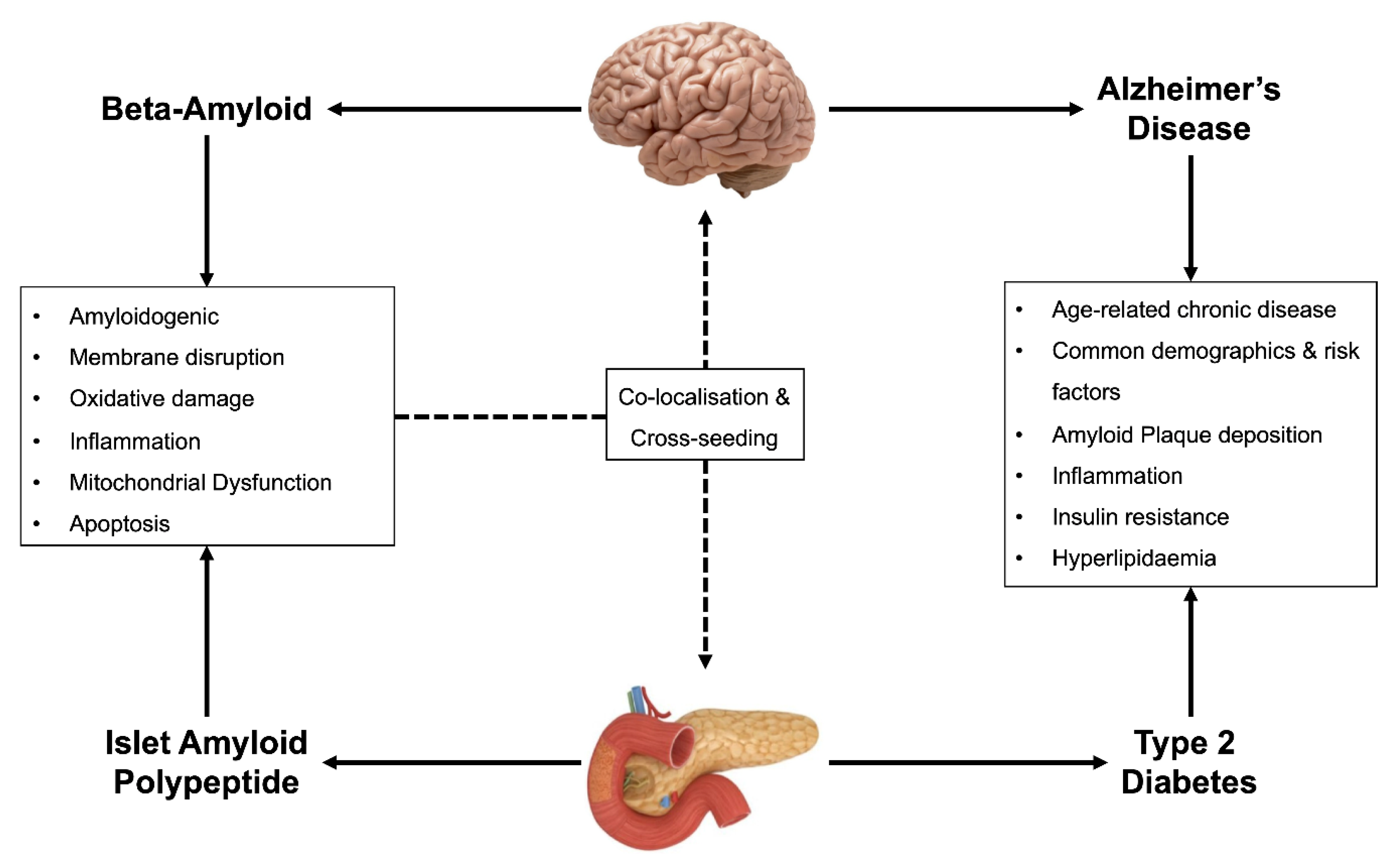
Insulin resistance is also a key feature of PCOS. Women with PCOS often struggle with metabolic issues, including difficulty regulating blood sugar, weight gain, and a highly increased risk of Type II Diabetes[4]. This overlap raises an important question: Could the metabolic dysfunction seen in PCOS increase the risk of Alzheimer’s disease in women?
Understanding PCOS
PCOS is a complex hormonal disorder that affects the ovaries and the body’s ability to regulate androgens. Common symptoms include irregular menstrual cycles, excess hair growth, acne, and ovarian cysts. PCOS also impacts metabolism, inflammation, and even brain health.
One of the noted features of PCOS is insulin resistance. Additionally, women with PCOS often have imbalances in estrogen and progesterone, hormones that play a protective role in brain health[4]. These hormonal and metabolic irregularities may potentially lead to the pathogenesis of neurodegenerative diseases like Alzheimer’s.
The Overlapping Science
The connection between Alzheimer’s and PCOS lies in their shared biological and pathological mechanisms. Both conditions are characterized by insulin resistance, chronic inflammation, and hormonal imbalances, all of which can negatively impact brain health.
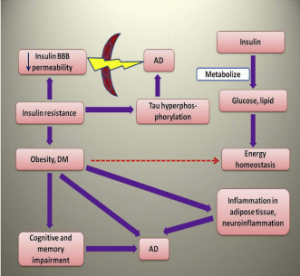
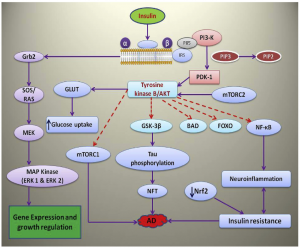
Insulin Resistance: When the body becomes resistant to insulin, it struggles to regulate blood sugar levels, leading to elevated insulin and glucose in the bloodstream. Insulin resistance is associated with increased production and reduced clearance of amyloid-beta plaques. Insulin-degrading enzyme helps to clear these plaques but is less effective when a person is insulin-resistant. Amyloid-beta plaques also bind to insulin receptors, thus further exacerbating insulin resistance. Insulin resistance can activate kinases that hyperphosphorylate tau proteins, causing them to form neurofibrillary tangles. These plaques and tangles are hallmarks of Alzheimer’s and cause cell death in the brain[3].
The relationship between PCOS and insulin resistance is complex and bidirectional, meaning they can influence each other in a cyclical manner. It’s not entirely clear which comes first, as both conditions are intertwined and can exacerbate one another. Elevated insulin levels directly affect the ovaries by producing more androgens and reducing the liver’s production of sex hormone-binding globulin, increasing the levels of free androgens in the bloodstream. Insulin resistance can also disrupt the hypothalamic-pituitary-ovarian axis, leading to hormonal imbalances that further exacerbate PCOS symptoms. In turn, hyperandrogenism from PCOS alters fat distribution and impairs glucose metabolism, causing insulin resistance[4].
Inflammation: Chronic, systemic inflammation is a key feature of both conditions, likely exacerbated by insulin resistance. In PCOS, inflammation is driven by metabolic dysfunction and elevated androgens. Studies have found increased levels of inflammatory markers, such as cytokines and c-reactive proteins, in women with PCOS[4], [5].
In Alzheimer’s, inflammation in the brain exacerbates neurodegeneration. As mentioned earlier, tau and amyloid-beta aggregation are hallmarks of Alzheimer’s. The presence of tau and amyloid-beta activates glial cells, which act as support and immune cells in the brain. These glial cells become chronically activated, which triggers the release of pro-inflammatory cytokines and reactive oxygen species[3]. The elevated levels of inflammatory markers in both PCOS and Alzheimer’s may suggest a shared inflammatory pathway.
Hormonal Imbalances: Estrogen, a hormone often imbalanced in PCOS, plays a protective role in brain health. It helps regulate glucose metabolism, reduces inflammation, and supports the growth and survival of neurons. Women with PCOS may experience fluctuations in estrogen levels, which could increase their vulnerability to Alzheimer’s, especially after menopause when estrogen levels decline[2].
Is PCOS a Risk Factor for Alzheimer’s?
While the exact relationship between PCOS and Alzheimer’s is currently being studied, there is growing evidence to suggest that women with PCOS may be at higher risk for cognitive decline later in life. A recent study found that women with PCOS performed worse on memory and cognitive tests compared to women without the condition, likely due to the increase in androgens[7]. Additionally, the metabolic and hormonal disruptions seen in PCOS (such as insulin resistance, inflammation, and hormone imbalance) are all known risk factors for Alzheimer’s[3]. While these studies begin to illustrate the relationship, future research should focus on the directionality of the links between insulin resistance, PCOS, and Alzheimer’s.
The connection between Alzheimer’s disease and Polycystic Ovarian Syndrome highlights the complex interplay between metabolism, hormones, and brain health. By understanding the shared mechanisms underlying these conditions (particularly insulin resistance and inflammation) we can develop more effective strategies for prevention and treatment. For example, metformin, a commonly used drug used to treat insulin resistance in women with PCOS, is being explored as a potential treatment for Alzheimer’s[3]. As research continues to uncover the links between PCOS and Alzheimer’s, it’s clear that addressing metabolic health is not just a matter of managing symptoms but a critical step in protecting women’s long-term brain health. “Type III Diabetes” serves as a powerful reminder that the health of the body and the brain are deeply interconnected and that women’s health deserves greater attention, investment, and education.
References
[1] M. M. Mielke, “Sex and Gender Differences in Alzheimer’s Disease Dementia,” Psychiatr. Times, vol. 35, no. 11, pp. 14–17, Nov. 2018.
[2] R. Deswal, V. Narwal, A. Dang, and C. S. Pundir, “The Prevalence of Polycystic Ovary Syndrome: A Brief Systematic Review,” J. Hum. Reprod. Sci., vol. 13, no. 4, pp. 261–271, 2020, doi: 10.4103/jhrs.JHRS_95_18.
[3] A. Akhtar and S. P. Sah, “Insulin signaling pathway and related molecules: Role in neurodegeneration and Alzheimer’s disease,” Neurochem. Int., vol. 135, p. 104707, May 2020, doi: 10.1016/j.neuint.2020.104707.
[4] A. Purwar and S. Nagpure, “Insulin Resistance in Polycystic Ovarian Syndrome,” Cureus, vol. 14, no. 10, p. e30351, doi: 10.7759/cureus.30351.
[5] S. Aboeldalyl, C. James, E. Seyam, E. M. Ibrahim, H. E.-D. Shawki, and S. Amer, “The Role of Chronic Inflammation in Polycystic Ovarian Syndrome—A Systematic Review and Meta-Analysis,” Int. J. Mol. Sci., vol. 22, no. 5, p. 2734, Mar. 2021, doi: 10.3390/ijms22052734.
[6] M. A. Rather et al., “Inflammation and Alzheimer’s Disease: Mechanisms and Therapeutic Implications by Natural Products,” Mediators Inflamm., vol. 2021, no. 1, p. 9982954, 2021, doi: 10.1155/2021/9982954.
[7] M. Perović, K. Wugalter, and G. Einstein, “Review of the effects of polycystic ovary syndrome on Cognition: Looking beyond the androgen hypothesis,” Front. Neuroendocrinol., vol. 67, p. 101038, Oct. 2022, doi: 10.1016/j.yfrne.2022.101038.










 [1]
[1] [5]
[5]