
Autism spectrum disorder (ASD) is an umbrella term for a developmental disability that can cause significant social, communication, and behavioral challenges. These individuals typically aren’t distinguishable by appearance but rather exhibit different methods of learning, interaction, and behavior. ASD is described as a spectrum since individuals can exhibit a diverse range of behaviors that vary in severity. High functioning autistic individuals may not be distinguishable from others, they can pursue education, earn a salary, and live independently while low functioning individuals may be nonverbal and require assistance.
ASD is a complex condition with many underlying conditions that influence and contribute to the risk of development. Challenges in studying ASD arise from the complex pathophysiology of the condition which involves neural connectivity, neural migration, excitatory and inhibitory imbalances, increased synapses, lack of mirroring in neurons, neuroimmunity disturbances, epigenetics, and dysfunctional genes, all of which have complex signaling and cross-communication of their own. To keep this article brief, we will only examine a couple of these to observe how they may impact the development of ASD.

Let’s begin by exploring imbalances in Glutamate (primary excitatory neurotransmitter) and GABA (primary inhibitory neurotransmitter). The Excitatory/Inhibitory balance between glutamate and GABA is critical for the proper functioning of neuronal networks and brain functions. It has been found that there are disturbances in glutamatergic and GABA-ergic receptors within ASD patients leading to an Excitatory/Inhibitory imbalance. This results in excessive glutamatergic excitation which may lead to excitotoxicity and cell death, as well as activation of glial cells that stimulate neurotransmission. To summarize, an imbalance in these important chemicals within the brain can lead to overstimulation of the brain and cause damage to neurons which ultimately results in reduced or altered brain function.
Another pathology relates to synaptic pruning. In neurotypical brains, the initial overabundance of dendritic spine formation is selectively pruned to support brain maturation, and this is regulated through a complex process abbreviated as mTOR regulated autophagy. In autism, there is a reduction in developmental dendritic spine pruning resulting in an impairment in mTOR regulated autophagy. Put simply, as we develop from infancy, our neurons make connections with one another some of which (in a healthy brain) are removed to reduce overstimulation and strengthen/reinforce the important pathways. In autistic individuals, this removal of unnecessary neural connections is not performed leading to an overabundance of connections and decreased efficiency in brain processing and communication.

Mirror neurons have also been found to contribute to the development of ASD. In a healthy brain, the mirror neuron system works to help with learning observed actions and emotions, but in autistic brains, fMRI have shown cortical thinning of regions belonging to the mirror neuron system. This means ASD symptom severity can be correlated to the limited development of the mirror mechanism which may appear in behavioral differences between autistic individuals and others.
Though there are several other contributions we could examine, the final component we will explore is genetic mutations which negatively impact synaptic transmission and synaptic growth. Dysregulation in gene networks forming both the synaptic transmission supporting system and the synaptic growth rate are implicated in the development of ASD, but no specific single gene modulation is sufficient to cause ASD symptom formation. This means more research is needed to elucidate the specific gene-environment & gene-gene interactions giving rise to ASD symptoms. This describes that genes play a significant role in the regulation of neural communication and that issues with this are found in patients with ASD. It’s also understood that it isn’t the dysfunction of a single gene that leads to the development of ASD but rather likely the contributions and combinations of multiple genes and uncovering these genes has yet to be done.

This was a brief overview exploring the very basics of a couple factors out of many that contribute to the complexities of understanding ASD. There remains much research to be done on the subject and a cure has yet to be discovered. Understanding the complex pathophysiology of this condition could provide insight into the dysfunction of countless other conditions and yield future treatment strategies.
Sources


 The mirror neuron system has a wide variety of functions when working normally. It develops in infants before they have reached 12 months of age and begins to let them understand others’ actions. As the famous Hebbian theory of neuroscience states, “cells that fire together, wire together”. This means that cells that are activated simultaneously form connections. This is also known as associative learning as it results in the brain forming an association between the two objects or concepts (for example, a glass of water and the feeling of thirst). Language development, which is in some ways a form of associative learning, also involved this system.
The mirror neuron system has a wide variety of functions when working normally. It develops in infants before they have reached 12 months of age and begins to let them understand others’ actions. As the famous Hebbian theory of neuroscience states, “cells that fire together, wire together”. This means that cells that are activated simultaneously form connections. This is also known as associative learning as it results in the brain forming an association between the two objects or concepts (for example, a glass of water and the feeling of thirst). Language development, which is in some ways a form of associative learning, also involved this system.



 unfortunately still be somewhat difficult to diagnose from a very young age, and so if we as humans have issues diagnosing it within ourselves, it could be even more difficult to actually pin down when observing an animal.
unfortunately still be somewhat difficult to diagnose from a very young age, and so if we as humans have issues diagnosing it within ourselves, it could be even more difficult to actually pin down when observing an animal.  discover similarities in a breed of dog called Bull Terriers, which they found to display some of the (dog equivalent) behaviors that can be diagnosed as autism in humans. I say “dog equivalent” because some of the staple behaviors of ASD are very difficult to distinguish in animals that can’t talk, such as imparied social interaction and not being able to communicate properly, which in humans can manifest as an inability to formulate speech or something similar.
discover similarities in a breed of dog called Bull Terriers, which they found to display some of the (dog equivalent) behaviors that can be diagnosed as autism in humans. I say “dog equivalent” because some of the staple behaviors of ASD are very difficult to distinguish in animals that can’t talk, such as imparied social interaction and not being able to communicate properly, which in humans can manifest as an inability to formulate speech or something similar.  just the behavioral aspect of autism with animals, but also potentially the physiological side of things too.
just the behavioral aspect of autism with animals, but also potentially the physiological side of things too. 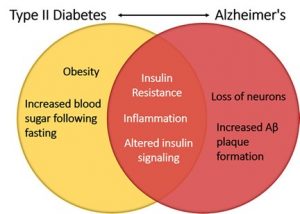
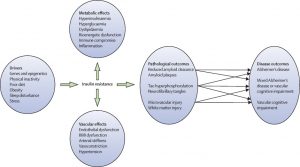


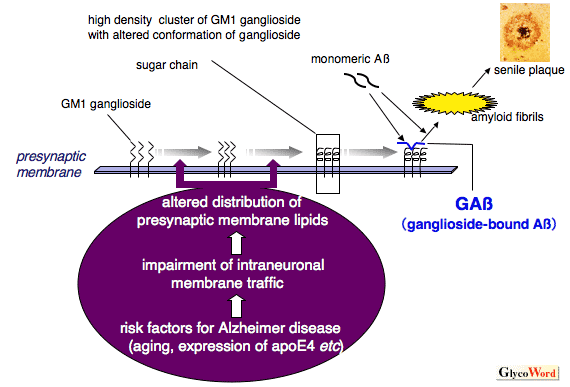
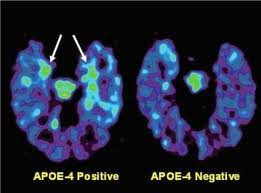


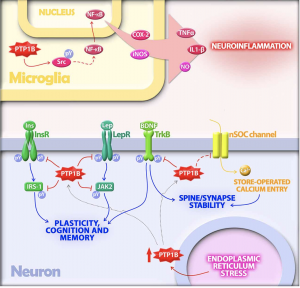
 potential treatment for both diseases, and luckily, scientists are working on this! Inhibiting PTP1B would result in no or less inhibition of insulin signaling, leptin receptors, and calcium channels even under endoplasmic reticulum stress. This would help to decrease the chance of developing type II in AD patients as well as decrease any neurodegenerative symptoms caused by PTP1B caused inhibition. But, it has been deemed an extremely difficult task due to the fact that the active site on PTP1B that an inhibitor would attached to is shared by over 100 other “family members” of this molecule. This makes it difficult for the inhibitor to only effect PTP1B and not any other PTPs present in other parts of the body.
potential treatment for both diseases, and luckily, scientists are working on this! Inhibiting PTP1B would result in no or less inhibition of insulin signaling, leptin receptors, and calcium channels even under endoplasmic reticulum stress. This would help to decrease the chance of developing type II in AD patients as well as decrease any neurodegenerative symptoms caused by PTP1B caused inhibition. But, it has been deemed an extremely difficult task due to the fact that the active site on PTP1B that an inhibitor would attached to is shared by over 100 other “family members” of this molecule. This makes it difficult for the inhibitor to only effect PTP1B and not any other PTPs present in other parts of the body.