Alzheimer’s is a complex disease that over time results in memory loss, confusion, and disorientation. This is why it is important to try to find either a preventative or treatment for Alzheimer’s. But in order to find a preventative or treatment, one must know what is going wrong in the brain causing this terrible disease to develop.
 In The Brain
In The Brain
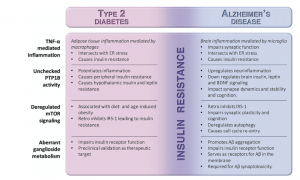
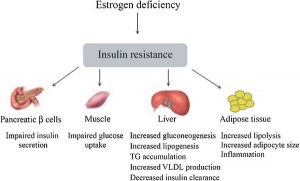
Although there are many things that are going wrong in the brain, the main cause of Alzheimer’s (AD) can be linked to one main issue: insulin resistance. Insulin resistance is the inability to produce or process insulin the way the body is supposed to. Insulin helps protect neurons, the brain’s cells that communicate with each other, and is important for learning and memory pathways. This means that insulin needs to be functioning properly in the brain, because when it’s not, there are detrimental effects. Most people think of Type 2 Diabetes (T2D) when hearing the word “insulin”. T2D is characterized by insulin resistance as well. This is ironic because T2D and AD have been linked to each other and both have common factors: inflammation and insulin resistance. There are four things that lead to insulin resistance: inflammation, too much PTP1B activity, not enough mTOR signaling, and abnormal ganglioside metabolism.
When insulin is functioning normally, it binds to insulin receptors located on the neuron. These receptors become activated and phosphorylate proteins called IRS1 and IRS2. Phosphorylation just means that phosphate molecules are put on the proteins, which then activates the proteins. These proteins then cause more activation and signaling resulting in insulin’s activities in the brain, like helping with learning and memory.
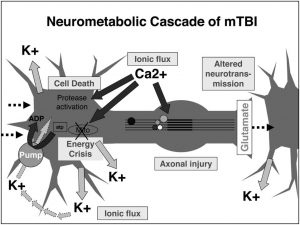
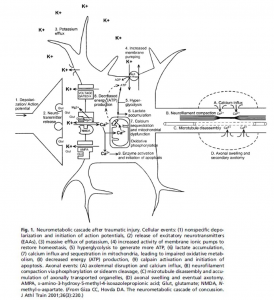
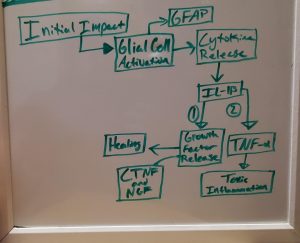
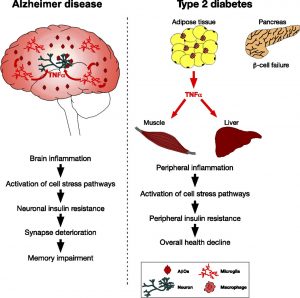
 However, like stated previously, in AD the insulin signaling is interrupted. This interruption happens because of activated, malfunctioning macrophages. Macrophages play an important role in the immune system and cell death. These macrophages secrete proinflammatory cytokines, which are chemicals that cause inflammation. One important proinflammatory cytokine in AD is called TNF-α. High concentrations of TNF-α cause significant inflammation, which interrupts the insulin signaling pathway, by preventing insulin receptors from activating IRS1 and IRS2 phosphorylation. Therefore, TNF-α causes insulin resistance, ultimately affecting learning and memory.
However, like stated previously, in AD the insulin signaling is interrupted. This interruption happens because of activated, malfunctioning macrophages. Macrophages play an important role in the immune system and cell death. These macrophages secrete proinflammatory cytokines, which are chemicals that cause inflammation. One important proinflammatory cytokine in AD is called TNF-α. High concentrations of TNF-α cause significant inflammation, which interrupts the insulin signaling pathway, by preventing insulin receptors from activating IRS1 and IRS2 phosphorylation. Therefore, TNF-α causes insulin resistance, ultimately affecting learning and memory.
Next, is malfunctioning PTP1B activity. PTP1B is an enzyme that regulates the insulin signaling pathway. It represses insulin normally, but when it is malfunctioning, it represses too much insulin. This happens because PTP1B dephosphorylates tyrosine residues and represses leptin. This is an issue because leptin is important for cognition and memory, as well as amyloid-beta protein plaque metabolism. This then turns off BDNF receptors causing insulin resistance and inflammation. Therefore, PTP1B just keeps dephosphorylating and turning off everything, causing insulin resistance and inflammation.
Third, deregulated mTOR signaling can cause insulin resistance. The mTOR signaling pathway regulates cell metabolism, proliferation, and survival. When mTOR signaling turns off, it also turns off insulin substrates that are no longer able to bind to their receptor. Ultimately, leading to insulin resistance. Lastly, ganglioside metabolism is malfunctioning in Alzheimer’s. The gangliosides contain a glycosphingolipid along with acids. They normally promote the accumulation of amyloid-beta protein plaques. But malfunctioning gangliosides improperly slice amyloid-beta precursor proteins (APP) which causes residues to clump together, promoting too much amyloid-beta protein plaques. Therefore, resulting in inflammation and AD.
 CBD Vs. THC?
CBD Vs. THC?
Before explaining CBD as a preventative or treatment for Alzheimer’s, it is important to note where it comes from and clear up any misconceptions that there might be about it. Cannabidiol (CBD) is a chemical found in the plant commonly known as marijuana or hemp. There are over 80 chemicals identified in this plant. One of which is the psychoactive chemical known as delta-9-tetrahydrocannabinol (THC). Like other chemicals in hemp, CBD can be extracted from hemp with little to no trace amounts of THC. Therefore, CBD does not get you high.
CBD as a Treatment?
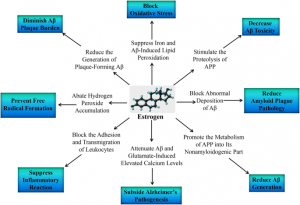
In a study, researchers found evidence that compounds found in marijuana, including CBD, can promote the cellular removal of amyloid beta. This study showed that cannabinoids affect both inflammation and amyloid beta accumulation in nerve cells. Although the studies were conducted on laboratory grown neurons, it still offers insight into the role of inflammation in Alzheimer’s disease. It also could provide clues to developing therapeutics for AD.
Another study had a control group and AD transgenic mice that were treated orally with CBD oil. The researchers then ran a series of tests before the cortical and hippocampal tissues in the brain were analyzed for amyloid-beta plaques, oxidative damage, cholesterol, phytosterols, and inflammation. The results showed CBD oil was able to prevent social recognition deficits and social withdrawal in mice with AD. Results also showed that CBD oil reversed cognitive deficits in mice with AD as well as provided neuroprotective, anti-oxidative, and anti-inflammatory properties. Therefore, this research provided evidence that CBD oil could potentially be used as a preventative for developing AD.
Lastly, there is evidence that CBD oil could be used to manage symptoms of AD by reducing oxygenation and inflammation, as well as stimulating and protecting the brain. CBD oil has also shown to reduce anxiety and stress that may trigger episodes of agitation and aggression in patients with AD. Though these studies have shown promising results for CBD oil being a preventative and treatment for AD. Before you take CBD oil, it is important to look at the potential side effects it may cause.
References
https://www.nature.com/articles/npjamd201612
https://content.iospress.com/articles/journal-of-alzheimers-disease/jad140921



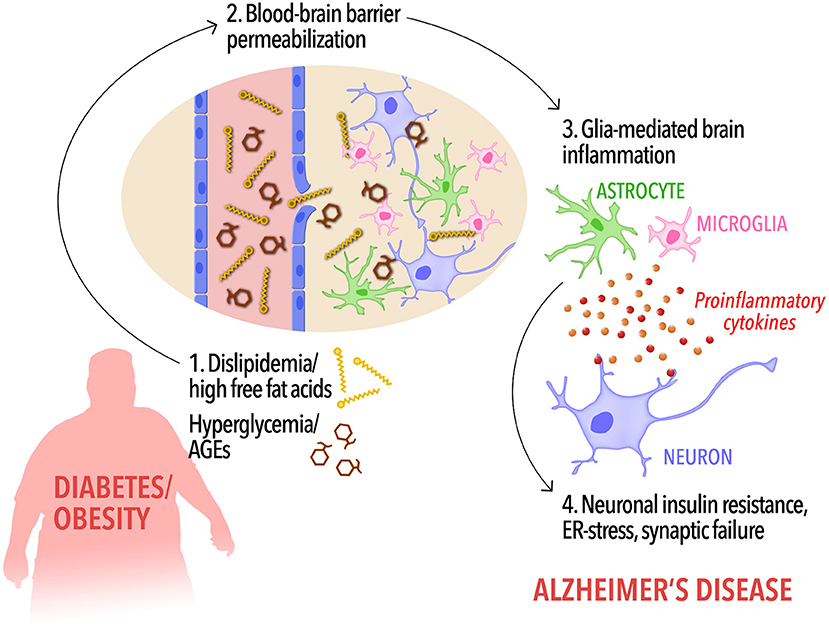



 When functioning normally, insulin in the brain binds to insulin receptors located on the neuron’s cell membrane. These receptors, once activated, phosphorylate proteins called IRS1 and IRS2, which means that phosphate molecules are put on the proteins. This in turn activates IRS1 and IRS2 which go off to cause more activation and signaling cascades resulting in insulin’s aforementioned activities in the brain, like facilitating learning and memory.
When functioning normally, insulin in the brain binds to insulin receptors located on the neuron’s cell membrane. These receptors, once activated, phosphorylate proteins called IRS1 and IRS2, which means that phosphate molecules are put on the proteins. This in turn activates IRS1 and IRS2 which go off to cause more activation and signaling cascades resulting in insulin’s aforementioned activities in the brain, like facilitating learning and memory. Definitely not! First of all, Infliximab is not a preventative drug meant to keep you from developing AD or T2D.
Definitely not! First of all, Infliximab is not a preventative drug meant to keep you from developing AD or T2D. 






 effects of rapamycin that was found was that, when intermittently administered, rapamycin led to stem cell regeneration. Considering some of the applications for stem cells, this is an amazing discovery that could have far reaching implications on the future of medicine. And that’s potentially just the tip of the iceberg. Because of the mTOR1 inhibition through rapamycin, it has been found that there is a decreased risk of cancer, since mTOR1 signaling can lead to cell proliferation. Tumor regression has also been observed to occur. It’s also been found that mTOR1 signalling sometimes leads to misfolded proteins. Using rapamycin, inhibiting mTOR1 led to greater autophagy and suppression of protein synthesis, which is important because this means that rapamycin can have neuroprotective effects in not just Alzheimers, but also Parkinson’s Disease and Huntington’s Disease.
effects of rapamycin that was found was that, when intermittently administered, rapamycin led to stem cell regeneration. Considering some of the applications for stem cells, this is an amazing discovery that could have far reaching implications on the future of medicine. And that’s potentially just the tip of the iceberg. Because of the mTOR1 inhibition through rapamycin, it has been found that there is a decreased risk of cancer, since mTOR1 signaling can lead to cell proliferation. Tumor regression has also been observed to occur. It’s also been found that mTOR1 signalling sometimes leads to misfolded proteins. Using rapamycin, inhibiting mTOR1 led to greater autophagy and suppression of protein synthesis, which is important because this means that rapamycin can have neuroprotective effects in not just Alzheimers, but also Parkinson’s Disease and Huntington’s Disease.  Before we all start taking insulin, it’s important to remember however that one of the most consequential aspects of a drug’s effects and effectiveness is the dosage and how often its administration. With rapamycin, the appropriate ratios of both of these factors are still unknown as of yet, but research is being done to try decipher further how to access the perceived positive effects of the drug. It has been seen though that rapamycin doesn’t fully inhibit mTORC1 processes, and so a combination with another treatment would more effectively implement the positive effects of Rapamycin.
Before we all start taking insulin, it’s important to remember however that one of the most consequential aspects of a drug’s effects and effectiveness is the dosage and how often its administration. With rapamycin, the appropriate ratios of both of these factors are still unknown as of yet, but research is being done to try decipher further how to access the perceived positive effects of the drug. It has been seen though that rapamycin doesn’t fully inhibit mTORC1 processes, and so a combination with another treatment would more effectively implement the positive effects of Rapamycin. increase insulin resistance, but the mechanism for why this occurs is speculated to be known (inhibition of mTOR2). Other negative effects included increased hyperglycemia within type 2 diabetes mouse models, and the ability for tumors to start regrowing after treatment with rapamycin stopped.
increase insulin resistance, but the mechanism for why this occurs is speculated to be known (inhibition of mTOR2). Other negative effects included increased hyperglycemia within type 2 diabetes mouse models, and the ability for tumors to start regrowing after treatment with rapamycin stopped.