
When we talk about obesity, we usually don’t instantly think about the brain. Primary areas of concern usually consist of the heart, the joints, and the metabolic system to name a few. However, some of the most consequential changes happen in the brain, specifically in a small but mighty region called the hypothalamus. Furthermore, emerging research suggests that these brain changes aren’t just a consequence of carrying extra weight – they’re actively driving it.
Inflammation at the Control Center
A 2017 review by Jais and Brüning in the Journal of Clinical Investigation laid out a striking picture: a high-fat diet doesn’t just expand the waistline: it triggers inflammation deep in the brain, and it does so fast. Within days of starting a high-fat diet, before significant weight gain even occurs, the hypothalamus begins showing signs of inflammatory stress. Key signaling pathways involving JNK and IKK activate, leptin and insulin resistance develops at the neuronal level, and the brain’s ability to accurately read the body’s energy state starts to break down. [1]
The hypothalamus is essentially the thermostat of your metabolism. It receives signals from hormones like leptin and insulin that tell it how much fat you’re carrying and how much you’ve eaten, then adjusts hunger and energy expenditure accordingly. When diet-induced inflammation disrupts this feedback loop, the thermostat breaks. You eat more, burn less, and the cycle deepens.
What makes this especially sobering is that it’s not just neurons involved. Microglia, astrocytes, and cells lining the blood-brain barrier all participate in — and amplify — this inflammatory cascade. Over time, chronic inflammation can even trigger the death of POMC neurons, the very cells responsible for suppressing appetite. The damage compounds quietly, long before it shows up on a scale.
The Good News: Some of This Is Reversible
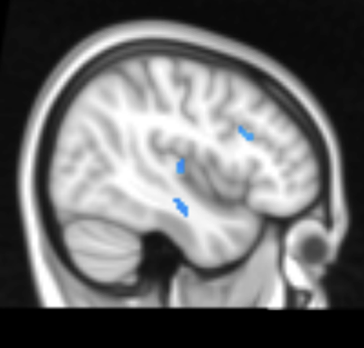
Here’s where things get more hopeful. Recent research into what happens to the brain after weight loss tells a more optimistic story. Following a structured weight loss intervention, activity decreased significantly in brain regions that are typically overactive in obesity – areas tied to food reward, taste processing, and decision-making around eating (figure 1). Critically, these changes didn’t show up after a 48-hour fast, meaning they weren’t just a side effect of short-term hunger. They appear to be genuine adaptations tied to actual reductions in body weight and fat mass. Leptin levels dropped alongside brain activity, suggesting the brain is recalibrating in response to a changed metabolic environment. [2]
The implications for eating behavior are significant. Less reactivity in reward-related brain regions could mean food simply becomes less compelling – less of a constant pull. Whether these changes help sustain weight loss long-term is still an open question, and researchers are careful to note the limitations of small study sizes.
Even more striking are findings from bariatric surgery research. Individuals with obesity showed measurably older brain ages compared to normal-weight peers – a marker of accelerated neurological aging. But after surgery and sustained weight loss, brain age improved by roughly three to six years over two years of follow-up. These weren’t subtle changes confined to one area; they were global, touching sensory, visual, and attention networks alike, and they correlated with lower BMI, better blood pressure, and improved insulin resistance. [3]
What This Means
Taken together, these findings reframe obesity not as a failure of willpower but as a condition that reshapes the very organ responsible for regulating appetite and behavior. The hypothalamus gets caught in a feedback loop where inflammation drives overeating, and overeating drives more inflammation. The brain, in a real sense, becomes organized around a higher weight set point.
But the brain is also adaptable. Weight loss – whether through lifestyle change or surgery – appears to walk some of those changes back. The thermostat can, at least partially, be reset. Understanding exactly how and why that happens may be one of the most important frontiers in obesity medicine.