Featured image created by Julia Wolf and Microsoft Copilot
The Science of Glioblastoma
Glioblastoma (GBM) is the most aggressive and deadly form of brain cancer. What makes it especially challenging isn’t just how fast it grows, but how complex it is. GBM tumors are highly heterogeneous, meaning no two tumor cells are exactly alike. This diversity allows the cancer to adapt quickly, resist treatment, and come back even after aggressive therapy [1].
Currently, standard treatment includes surgery, radiation, and chemotherapy. Even with all three, the average survival time is only about 14 months, and long-term survival is rare. One major reason for this is that GBM cells don’t stay in one place. They invade nearby brain tissue, making them nearly impossible to remove them completely through surgery. Even a few remaining cells can regrow a new tumor. Another key challenge is something called the blood-brain barrier (BBB). This protective barrier helps keep harmful substances out of the brain, but it also blocks many cancer drugs from reaching the tumor effectively. As a result, treatments that work well for other cancers often fail in GBM [1].
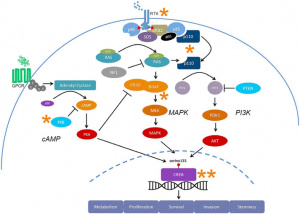
On a deeper level, GBM is driven by complex communication systems inside cells known as signaling pathways. Three major ones, PI3K, MAPK, and cAMP, control how cells grow, survive, and spread. In glioblastoma, these pathways are often overactive or disrupted, pushing cells to divide uncontrollably and resist death . Even more challenging, these pathways don’t act alone, they interact and “talk” to each other, creating backup systems that help the tumor survive even when one pathway is targeted. Because of this, researchers now believe that treating GBM effectively will require targeting multiple pathways at once—or finding ways to interrupt key “hubs” where these signals converge [1].
To learn more about the science of glioblastoma, click here!
Immunotherapy: Turning the Body Against Cancer
One of the most exciting features of immunotherapy is something called immunological memory. In some cases, the immune system can “remember” cancer cells and continue to fight them long after treatment, reducing the chance of recurrence. However, like all treatments, immunotherapy comes with risks. Side effects vary widely, from mild flu-like symptoms to more serious immune-related conditions that resemble autoimmune diseases [2].
Types of Immunotherapy and Their Role in GBM
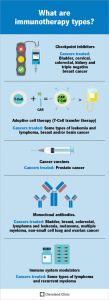
Immune Checkpoint Inhibitors (ICIs)
Our immune system has built-in “brakes” (like PD-1 and CTLA-4 receptors) that prevent it from attacking healthy cells. Cancer cells often exploit these checkpoints to hide from immune attack. Checkpoint inhibitors block these brakes, allowing T-cells to stay active and attack tumors. These therapies have been highly successful in cancers like melanoma. For glioblastoma, however, results have been more limited. Drugs like nivolumab and pembrolizumab have shown only modest improvements in survival, likely due to the tumor’s complexity and its ability to suppress immune responses [3].
Cancer Vaccines
Cancer vaccines aim to train the immune system to recognize specific cancer-related markers. Some are preventive (like the HPV vaccine), while others are therapeutic [4]. In GBM, results have been mixed, but certain approaches, like dendritic cell vaccines (e.g., DCVax-L), have shown promise in improving survival in some patients [5].
Adoptive Cell Therapy and CAR T-Cells
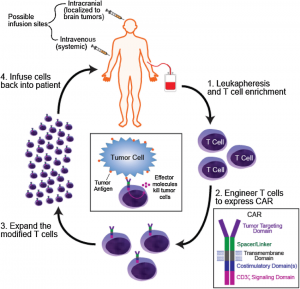
Doctors collect a patient’s immune cells, reprogram them to better recognize cancer, grow many more of these enhanced cells, and then return them to the body so they can seek out and destroy tumor cells [7].
This is one of the most advanced and personalized forms of immunotherapy. T-cells are taken from a patient, modified in a lab, and then returned to the body to attack cancer. A powerful version of this is CAR T-cell therapy, where T-cells are engineered to recognize specific proteins on tumor cells. In glioblastoma, CAR T-cells are being designed to target proteins like IL13Ra2, HER2, and EGFRvIII. Researchers are even developing “trivalent” CAR T-cells that can target multiple markers at once to overcome tumor diversity and prevent resistance. Because of the blood-brain barrier, these cells are often delivered directly into the brain or spinal fluid rather than through the bloodstream [6].
The Future: Combining Strategies and Why This Matters
The future of glioblastoma treatment likely won’t rely on a single solution. Instead, it may involve multi-layered approaches, combining:
- Targeted therapies (blocking signaling pathways)
- Immunotherapy (activating the immune system)
- Traditional treatments (surgery, radiation, chemotherapy)
By attacking the tumor from multiple angles, scientists hope to overcome its adaptability and improve long-term outcomes.
Why this matters: GBM is so difficult to treat because it constantly changes and resists single therapies. A combined research approach could not only slow tumor growth but also extend survival, improve quality of life, and move treatment closer to long-term control, or even a cure.
References
[1] N. H. Fung et al., “Understanding and exploiting cell signalling convergence nodes and pathway cross-talk in malignant brain cancer,” Cellular Signalling, vol. 57, pp. 2–9, Jan. 2019, doi: https://doi.org/10.1016/j.cellsig.2019.01.011.
[2] Cancer Research Institute, “What is Immunotherapy?,” Cancer Research Institute, 2023. https://www.cancerresearch.org/what-is-immunotherapy
[3] Z. Sarfraz, A. Maharaj, Vyshak Alva Venur, J. D. Lathia, Yazmin Odia, and M. S. Ahluwalia, “Immunotherapy in Glioblastoma: An Overview of Current Status,” Clinical Pharmacology Advances and Applications, vol. Volume 17, pp. 185–209, Jul. 2025, doi: https://doi.org/10.2147/cpaa.s497903.
[4] Cleveland Clinic, “Immunotherapy: Cancer Treatment, CAR T-Cell Therapy, Types, Risks,” Cleveland Clinic, Oct. 01, 2020. https://my.clevelandclinic.org/health/treatments/11582-immunotherapy
[5] L. Gatto, Vincenzo Di Nunno, A. Tosoni, S. Bartolini, L. Ranieri, and E. Franceschi, “DCVax-L Vaccination in Patients with Glioblastoma: Real Promise or Negative Trial? The Debate Is Open,” Cancers, vol. 15, no. 12, pp. 3251–3251, Jun. 2023, doi: https://doi.org/10.3390/cancers15123251.
[6] A. S. Luksik, E. Yazigi, P. Shah, and C. M. Jackson, “CAR T Cell Therapy in Glioblastoma: Overcoming Challenges Related to Antigen Expression,” Cancers, vol. 15, no. 5, p. 1414, Feb. 2023, doi: https://doi.org/10.3390/cancers15051414.
[7] Schematic of car T cell therapy for GBM | Download Scientific diagram, https://www.researchgate.net/figure/Schematic-of-CAR-T-cell-therapy-for-GBM_fig1_355420545 (accessed Apr. 24, 2026).