Throughout our media, we see headlines about obesity and the plague-like effect it has over our country. Shows and lifestyle programs are dedicated to stopping obesity. Michele Obama advocated for the elimination of obesity in schools. Messages infiltrate our TVs and radios that tell us to eat less processed food and exercise daily to combat obesity. However, is it possible to avoid or eliminate obesity by simply changing our diet and lifestyle? Or is there more to the problem… Is obesity an actual disease that must be treated with more finesse?
Obesity is indeed an actual disease and despite contrary belief, is not based on laziness or lack of self-control.
In a normal functioning brain, when we consume food, insulin and leptin proteins activate a very specific pathway that leads to ion efflux from specialized brain cells called neurons. This ion efflux of potassium leads to an action called hyperpolarization. Hyperpolarization is important because it slows the neuron firing that tells us to eat more. There are two types of neurons that control our eating: POMC-neurons, which tell us to stop eating, and AgRP-neurons, which tell us to eat more. Insulin acts on these neurons by decreasing the number of AgRP-neurons in the brain and increasing POMC-neurons. Therefore, when we eat and our body releases insulin, we respond by stopping our eating. However, in people who suffer from obesity, overnutrition occurs and normal cellular function becomes cellular stress. Specifically, ER stress leads to physical inflammation of the brain and this inflammation creates insulin resistance. Therefore, the insulin in the brain of someone suffering from obesity does not function properly and cannot tell the body to stop obtaining food. Overall, the effect of overnutrition and obesity becomes a deadly cycle of continual eating and weight gain.
The biological cycle of obesity occurring in the brain cannot be fixed by exercise or dietary changes alone. Drugs that can block the pathway or stimulate normal pathways are more likely to create brain reversals that have a positive effect.
So, the next time we see a show or program about obesity and we are quick to judge the lack of self-control, let us remember that obesity is a disease that physically effects the brain and requires medication or surgery in most cases. It is not a matter of laziness, but of better treatment options and open-mindedness.
http://www.cell.com/trends/endocrinology-metabolism/pdf/S1043-2760(12)00204-4.pdf
Too Much Fat in Your Diet?
From being too busy to simply being lazy, it is really easy to decide against cooking breakfast, lunch, or dinner and going out to eat instead. With McDonalds and Taco Bell being on every street corner, they are easier accessible and family cheap, this makes it way more tempting to eat out than to cook a nice healthy meal at home. On occasion this is perfectly fine, however overtime this can add up and cause all sorts of problems within the body. In today’s society, there is stress beyond belief coming from work, school, family, and friends, which pushes people to eat those unhealthy meals as well. These high fat foods make the body feel temporarily satisfied, which is rewarding and will condition people to continue to eat these foods.

So What is Happening in Your Body?
After having a high fat diet for a prolonged period of time, even after just 72 hours, the body starts to experience something called hyperinsulinemia. This is when there is an excess amount of insulin floating throughout the blood stream. This leads to stress within the brain, more specifically within the endoplasmic reticulum. Due to this ER stress, a couple of different pathways called Socs3 and JNK are activated which, in the end, leads to insulin resistance. Insulin resistance is when insulin produced within the body functions improperly and does not break down glucose. Since glucose is no longer being broken down as frequently as it should, this leads to high blood sugar levels which is a sign of type two diabetes. This process does not happen overnight, and can take years and years of a high fat diet for glucose levels to reach levels of being diabetic. So if you are worried about this happening, chances are its not too late to turn your diet around and reverse these changes.

Type Two Diabetes
Type two diabetes is diagnosed through a series of blood tests. These blood tests simply look at the levels of glucose within the blood and once it reaches a certain point a diagnosis of pre-diabetic or diabetic will be placed on the individual. Some of the symptoms of type two diabetes are increased thirst and urination, increased hunger, numbness or tingling in hands and feet, unexplained weight loss, and having sores that don’t heal properly. The main causes of type two diabetes are being overweight and obese, insulin resistance, the absence of physical activity, and genes. Since this is a genetic form of diabetes, it is possible to be diagnose with it, without being overweight or obese.
Sources:
https://moodle.cord.edu/pluginfile.php/625315/mod_resource/content/0/2013%20CNS%20insulin.pdf
The Science Behind Lou Gehrig’s Disease
Amyotrophic Lateral Sclerosis (ALS) is more commonly known as Lou Gehrig’s disease after the New York Yankee baseball player Lou Gehrig. During his season in 1938, Lou Gehrig started to experience muscle weakness which started to affect his batting and even his ability to tie his shoes. A diagnosis of ALS was soon made, bringing this otherwise not well known disease into the public’s eye.
ALS is characterized by muscle weakness or cramps, cognitive impairments, slurred speech, or difficulty swallowing. The disease often appears with similar symptoms to those of Parkinson’s disease or even Alzheimer’s disease, making the diagnosis of this disease based mainly on ruling out other diseases.
ALS is a degenerative disease that causes death in motor neurons of the brain, spinal cord, and nerves running to muscles. Motor neurons signal movement to occur in muscles by sending a signal from the brain to peripheral muscles. Death to neurons is caused genetics and oxidative stress on the cell.
One of the most important ideas to understanding the science behind ALS is to understand the role of the mitochondria in the cell. The mitochondria are the powerhouse of the cell. It helps to break down molecules so that the cell can use them for energy.
When the mitochondria become overworked, this leads to mitochondrial stress. During mitochondrial stress, extra molecules called oxidants are produced that are harmful to the cell. In response to excessive amounts of oxidants, the cell turns on pathways that signal the cell to die.

Oxidants are the cause of motor neuron death.
There are a few ways to get rid of oxidants. The way that the body normally uses is the SOD1 protein which breaks down oxidants into less harmful substances. Another way is by activation of the OXR1 gene which is the bodies natural response to oxidants. The last way to get rid of oxidants is through antioxidants which protect the mitochondria against oxidative stress.
All of these methods of getting rid of oxidants are currently being used in research to try to slow down the progression of ALS. There are currently no existing treatments for ALS that target oxidants as it can be very hard to target the exact motor neurons where the extra oxidants are forming. Future research will continue to look into oxidants as well as stem cells, optogenetics, and other medications.
Although Lou Gehrig might have been one of the first public cases of ALS, there have been many more cases and much more research conducted since his death. Lou Gehrig died from ALS, but his advocacy for the disease has lead to increased amounts of research, and promise for more effective treatment around ALS.
Information from this post can be found here
To find out more about ALS click here
More information about Lou Gehrig can be found here
Picture is original art work by the author
Obesity – More Than Self-Control?
Obesity is sometimes viewed as a lack of discipline. The obvious fix is start eating healthier and work out a few times a week. Although eating right and exercising are very important, obesity is more than just a weight problem.
There are neurological components to this disease that is plaguing the United States. One component of this very complex illness is the dysregulations of signals being sent in one’s brain.
In the hypothalamus and brain stem, there are specialized neurons called proopiomelanocortin neurons or POMC neurons for short. These neurons are vital players in the story of food control. POMC neurons send projections throughout the brain that let you know you’re full and should stop eating. The POMC works in tandem with a peptide called the agouti-related peptide (AgRP). This peptide is responsible for the signal to eat.
One regulator of POMC is a hormone called leptin. Leptin flows through the blood and when it reaches the brain it binds to the POMC neurons, therefore sending a signal to stop eating. When this process is interrupted in rats, obesity is seen as a result. Other mutations to POMC can lead to adrenal failure, along with early on-set obesity. Therefore, it is evident that this process is vital for maintaining a healthy weight.
This result of over-eating can further damage the body by leading to insulin-resistance. When over nutrition occurs, it can lead to ER stress which activates a pathway called the JNK pathway. This pathway is an inflammatory pathway which ultimately ends in inhibiting the insulin receptor substrates. This inhibition therefore leads to insulin resistance as the insulin signal will no longer be able to be passed on down it’s cascade of signals.
Obesity is large problem in our country. This problem comes with a stigma that those individuals who are obese are weak and have control problems. However, when further examining the components of this disease there are many neurological components. It is an imbalance of chemicals and other peptides. Therefore, I believe that with more research obesity will shift to being seen as a brain disease as well, hopefully improving the treatment and reducing the incidence.
For more reading on obesity and the brain, please check out:
http://www.cell.com/trends/endocrinology-metabolism/fulltext/S1043-2760(12)00204-4
ALS and Oxidative Stress – A Possible Culprit
ALS is a complicated and sinister illness. Over time it deteriorates the motor neurons in your brain and spinal cord, trapping its victims inside of their own bodies. Researchers are looking into how oxidative stress plays a role into causing this heartbreaking disease.
Oxidative stress is a result of the mitochondria, the organelle that makes energy in the cell, not being able to keep up with detoxifying the hazardous reactive products of oxygen. An increase in oxidative stress is considered to play a large role in the development of ALS.
When oxidative stress occurs it causes proteins to gather in the cytoplasm, which is where they are not suppose to be. This new aggregation of proteins combines with naturally occurring stress granules. For short periods of times, stress granules are beneficial. However, when present for long periods of time the beneficial effects turn into hazardous effects.
These harmful bundles in the cytoplasm can lead to misfolded proteins. The body has it’s own safety guard to protect against misfolded proteins. This safety guard is called the unfolded protein response (UPS).
https://www.nature.com/articles/nm0410-396
The unfolded protein response is the cell’s response to proteins that are made incorrectly. This response monitors the amount of proteins that are defective and when the number of defective proteins are too high it halts the production of proteins. This stop in protein making is done in order to correct the current mistakes. However, if this process takes to long the goal of the UPS switches form restoration to destruction causing the cells to initiate apoptosis, or cell death.
Therefore, increased and decreased amounts of UPS have been seen in ALS patients. With a low UPS there are many unfolded and misfolded proteins in the body not behaving as they should be, and with the upregulation of UPS many cells are dying.
Oxidative stress seems to be one of the culprits for initiating this cascade of negative effects and should continue to the a main point of focus in future research. With more research and understanding, hopefully one day a more curative treatment will be developed.
For more reading on the topic, please check out:
https://www.ncbi.nlm.nih.gov/pubmed/27150074
ALS: Holding on to Hope
One of the greatest blessings we often take for granted is our privilege to be able-minded and able-bodied. We live our lives boldly, making life goals and holding expectations that often rely on our current functionality. The reality is that one moment can change everything.

Unfortunately for some, that moment is their diagnosis with a motor neuron disease known as amyotrophic lateral sclerosis (ALS). This disease attacks motor neurons that control your muscle movement, which can progressively affect your ability to run, walk, stand, talk, and eventually breathe.
At a neurochemical level, genetic mutations have caused changes in RNA and the mitochondria. As of now, researchers know that oxidative stress is damaging mitochondria and causing RNA dysfunction, as RNA-binding proteins (FUS and TDP43) are being spliced and aggregated within the cytoplasm.
However, this RNA dysfunction is also contributing to the oxidative stress damaging mitochondria. FUS and TDP43 are reducing protection against oxidative stress and upregulating the enzymes responsible for increasing the levels of oxidative stress.
These mutated proteins contribute to misfolded proteins, leading to defective organelles and cell death. Aside from RNA mutations, researchers have also considered the role of iron and copper metabolism and their role in reducing oxidative stress.
As FUS and TDP43 are accumulating, the motor neuron axons begin to retract because the transporter proteins are not efficiently carrying information down the axon to trigger muscle contraction. As these neurons become less able to communicate their information, the cells become smaller and sometimes die. This cell death is what leads to the muscle atrophy and paralysis commonly associated with ALS.

An ALS diagnosis may bring a feeling of hopelessness. Without previous knowledge or signs of its onset until it has fully progressed, one can feel trapped within their own body. Currently, there is no cure; only treatment to slow its progression. As a society, we need to support those individuals by funneling money toward research to understand the progression of this disease. With these answers, we can seek better treatment and restore hope for those affected by ALS.
Steven Hawking, one of the most insightful intellectuals of our time, expands on his freedom to be able-minded, even though his ALS stripped him of being able-bodied. While most would rightfully feel the burden of the motor neuron disease, Steven’s slow-progressing form of the disease has given him time to reflect on the freedom of being able-minded.

He has proven through his contributions to science and philosophy that life satisfaction is based on perspective: one can feel trapped by the disease, or to use that life-changing diagnosis to redirect their path with the time they have left. I will leave you with my favorite quote of Steven’s:
“There should be no boundaries to human endeavor. We are all different. However bad life may seem there is always something you can do, and succeed at. While there is life, there is hope.”
The Complications of ALS
ALS is not a very common disease, probably due to its complexity, but has affected many lives nonetheless over the years. Families have seen their kin come and go as the disease vastly destroys their system. It is hard to see people go as fast as they do with ALS (usually only living between 2-3 years after diagnosis) and it is no surprise why after looking at the biology of things.
ALS begins with muscle weakness, stiffness, tightness, or spastic activity, which is occurring due to the breaking down and dying of motor neurons. When diagnosed, signs of upper and lower motor neuron degeneration begin to show, and an MRI is used to follow the progression of the disease. There are two types of ALS: Sporadic and familial. The sporadic form of ALS makes up roughly 90% of all cases, and is caused randomly. The familial form for ALS is passed on genetically, usually with a 50% chance. A reason that sporadic may be more prevalent than familial is the fact that after diagnosis, not many people try to start a family life, and those that are diagnosed and pass on the trait, probably already had families before diagnosis.

ALS is caused by a build up of reactive oxygen species (ROS) in the neuron, which ends up killing the neuron due to its toxicity. The reason for this buildup is due to several genetic mutations within the system. The most common of mutations resides on TDP43, which regulates RNA splicing. this mutation is in roughly 97% of all ALS cases, and is also found in frontotemporal dementia patients (FTD). This might explain that when the disease progresses, it no longer only affects motor neurons, but neurons involved with cognition and memory as well.
ALS has recently been focused on more and paid attention to due to the popularity of the ALS ice bucket challenge. In this challenge, a bucket of ice water is simply poured over each contestant, and then the contestant challenges another individual, or group of individuals, to do the same. ALS’s popularity has since grown, and funding has been on the rise. The lack of funding that has been shown for research in the past is probably due to the lack of affected population for the disease (2 cases per 100,000 people). Since then, situational awareness for ALS has significantly improved, and diagnosed individuals are living longer and healthier lives.

It is sad to see such a harsh disease affect people that we know and love, but it is really inspiring to see other individuals step up and support, especially when they have not been affected at all. Although research in the field of ALS is difficult, due to it being genetically based, I believe that the surge in research will at the very least provide better care for diagnosed individuals and prolong their life, while also driving to find a cure.
RESOURCES:
https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Amyotrophic-Lateral-Sclerosis-ALS-Fact-Sheet%20
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4404432/pdf/13311_2015_Article_338.pdf
Struggling to Hold On: The ALS Story
You’re Not YOU
Picture this: You are 29 years old, in shape, happily married, thinking of children, an avid baker, and enjoying playing the piano in the evenings as you sip your daily glass of red wine. Could life get any better?
Suddenly, one day you start to notice unusual, yet slight tremors in your fingers. You don’t think anything of it. But then those tremors get larger, more pronounced, enough to disrupt your usual evening piano recital and eject the glass of red wine from your hand. Welcome to the beginning of one of the most relentless and soul-robbing diseases today: Amyotrophic Lateral Sclerosis (ALS) or Lou Gehrig’s Disease.
Meet Kate, a classical pianist who is diagnosed with ALS and the star of the movie shown below, “You’re Not You.” Throughout the span of a few years, Kate experiences the progressive weakening and atrophy of her voluntary muscles that rob her of the life she loved to live. If you would like to experience the life of an ALS victim through the eyes of Hollywood drama, spend your Saturday afternoon sobbing and watching “You’re Not You.”
What is ALS?
ALS is a fatal motor neuron disease that is characterized by the weakening and atrophy of both upper and lower motor neurons. Normally upper motor neurons in the brain function to send signals to facial and cranial muscles, as well as to lower motor neurons located in the brain stem and spinal cord. The lower motor neurons then send signals out to respective muscles and cause contraction of those muscles to aid in our voluntary movements, such as talking, swallowing, breathing, and walking.
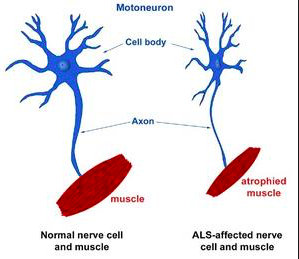 Figure 1. Depiction of a healthy motor neuron and an atrophied motor neuron seen in ALS.
Figure 1. Depiction of a healthy motor neuron and an atrophied motor neuron seen in ALS.
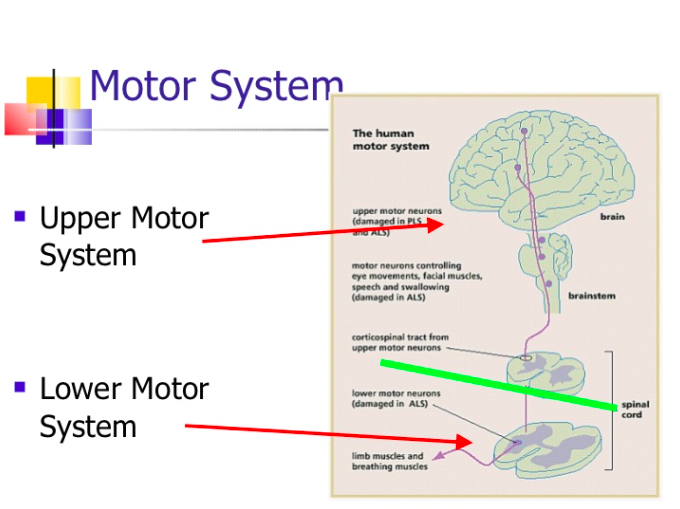 Figure 2. General locations of the upper and lower motor systems in the body.
Figure 2. General locations of the upper and lower motor systems in the body.
Trapped
Although all voluntary muscles are affected by ALS, the cognitive state of victims usually remains in tact. Some would argue that this makes ALS even more torturous for its victims to suffer through. This relentless disease almost always results in respiratory failure and death within a few short years (3-5 years) after initial diagnosis.
The ALS Brain
There are four interconnected pathways identified in the vicious cycle and progression of ALS that specifically affect both the upper and lower motor neurons of ALS victims. Researchers are currently unsure which part(s) of these four pathways is responsible for initiating this vicious cycle. These pathway outcomes ultimately lead to the associated symptoms of ALS:
1. SOD1 and Oxidative Stress
Many ALS cases involve mutations (genetically inherited or sporadic mutation) of the gene encoding the superoxide dismutase enzyme (SOD1). SOD1 functions as a cytosolic antioxidant enzyme, defending against reactive oxygen species (ROS) formed in cells during normal cellular metabolism. When the SOD1 gene is mutated, SOD1 enzyme is defective, which leads to a buildup of ROS in the brain and ultimately oxidative stress. The oxidative stress overpowers cellular organelles and leads to organelle dysfunction. The cellular organelle dysfunction leads to defective cellular metabolism and protein production and a prolonging of cellular oxidative stress.
2. Protein Folding
An increase in oxidative stress leads to the aggregation of misfolded proteins, which overpowers the body’s natural autophagy system (responsible for cleaning up misfolded proteins and defective organelles) and renders it dysfunctional. This amplifies the buildup of misfolded proteins in cells.
3. RNA Dysfunction
When under stress, the cell halts translation via the formation of stress granules in the cytoplasm: globules of non-translating mRNA, ribosomal subunits, and translation initiation factors (think of wads of gum). During prolonged oxidative stress, however, permanent stress granules form that cause important molecules in RNA processing and protein production (FUS and TDP43) to aggregate and render them nonfunctional. The stress granules are similar to wads of gum that stick to anything they touch, including FUS and TDP43. The failure of FUS and TDP43 to carry out RNA processing leads to dysfunctional RNA processing and protein production. This further amplifies cellular oxidative stress.
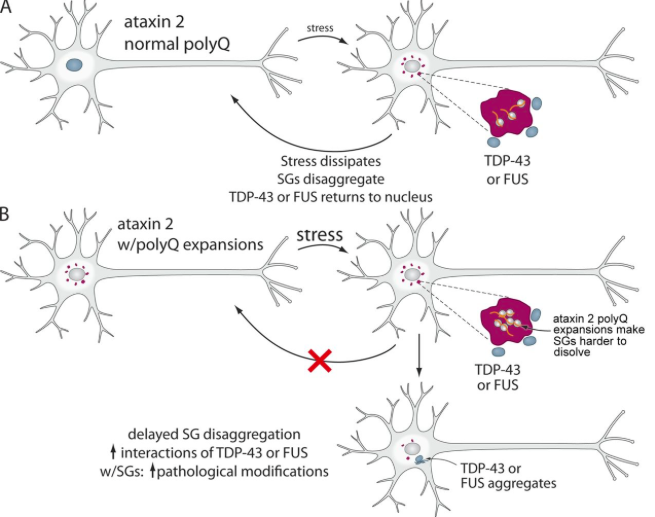 Figure 3. The formation of permanent stress granules in the cytoplasm and consequent aggregation of TDP43 and FUS.
Figure 3. The formation of permanent stress granules in the cytoplasm and consequent aggregation of TDP43 and FUS.
4. Metal Ion Homeostasis
An altered metabolism and increase in cellular levels of iron and copper lead to an increase in cellular ROS. This increases the oxidative stress in the body, amplifying the dysfunctional RNA processing mentioned before, and ultimately protein degradation within the cells.
The Future of ALS
Currently, there is no cure for this horrible, life-altering disease. However, there are multiple therapeutic options (physical therapy, occupational therapy, rehabilitation) to help ALS victims. There are also standard pharmaceuticals administered for pain relief and to aid in prolonging life, such as edaravone. Riluzole is a drug that has been shown to prolong the life of an ALS victim 2-3 months without treating symptoms of the disease.
The future of ALS lies in the hands of scientific researchers and the study of brains of ALS victims to develop a better understanding of the disease’s actions. Further pathways of the disease need to be identified in order to find potential therapeutic targets that may lead to the cure of ALS or a greater prolonging of life of ALS victims.
If you would like to learn more about the physiology of ALS, please visit:
http://www.sciencedirect.com/science/article/pii/S0304394016302877?via%3Dihub
Images From:
https://kin450-neurophysiology.wikispaces.com/ALS+II
https://www.slideshare.net/santhurao/physiology-859392
http://jcb.rupress.org/content/201/3/361
What We Don’t Know About ALS
It all starts out with muscle weakness in either the hands or the feet, and slowly progresses to slurred speech, stumbling, bad posture. These are just some of the early symptoms of ALS and they only get worse as the disease progresses. Which, in most cases, is a relatively fast progression. In most cases of ALS, the individual diagnosed will generally pass away from the disease within 3 years. However, there are cases of people that live 13 or 14 years, maybe even more with the disease but as sad as it is, these are rare cases.

Forms of ALS
There are two different forms of ALS, the first form is sporadic ALS.
Sporadic ALS makes up roughly 90% of all cases and is defined as a random onset of the disease. There is not a genetic factor to this form, meaning it is not passed down from family member to family member. However, family members of those diagnosed with sporadic ALS are at an increased risk of being diagnosed themselves, this is still a relatively low risk.
The second form of ALS is familial.
Familial ALS is just as it sounds. The genetic form, meaning it is passed down from family member to family member. This makes up only 10 % of all cases. Within this 10% there is roughly a 25-40% chance ALS is caused by a mutation on the C9orf72 gene. This gene mutation is also associated with atrophy in the frontal lobe of the brain, which is linked to frontal lobe dementia. This form of dementia could possibly be associated with the cognitive declines that go along with ALS.
So What is Causing ALS?
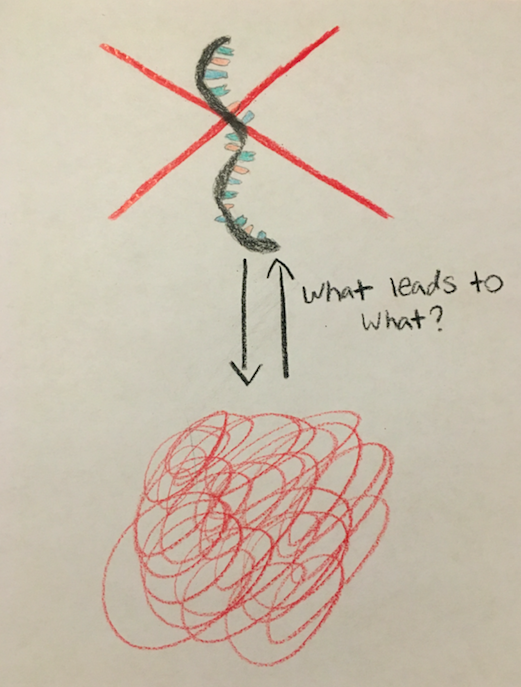
There are two main ideas about the cause of ALS, however it is unknown which one comes first in the pathway.
It is believed that oxidative stress within the brain leads to RNA dysfunction, which is linked to the onset of ALS. It is also believed that RNA dysfunction leads to oxidative stress which then leads to the onset of ALS. This is like a vicious cycle because we don’t know what causes what. Does RNA dysfunction cause oxidative stress or does oxidative stress cause RNA dysfunction? We are also unsure of what is causing either of them to occur in the first place, which is what science is trying to figure out.
It IS known that the proteins TDP43 and FUS are spliced away from the nucleus and aggregate in the cytoplasm. They are then localized and trapped within stress granules, and this is somehow leading to the onset of ALS.
Sources:
https://www.webmd.com/brain/understanding-als-symptoms#1
https://moodle.cord.edu/pluginfile.php/625305/mod_resource/content/0/ox%20stress%20and%20mito%20damage%20in%20ALS%202016.pdf
ALS, It’s More Than the Ice Bucket Challenge
Remember a few years back when the ice bucket challenge was a big thing? Everyone was doing it and the challenge went viral. Next thing I know my antisocial media mother is doing it too. She decided to create a Facebook just so she can get a bucket of ice cold water dumped on her. She didn’t do it just because all her friends were doing it or to be cool (maybe?). She did it to spread awareness about a rear and fetal disease called ALS. At first I didn’t know what ALS was or why it was taking over the world. But as time passed, I became aware of the disease and how prevalent it was.
So what is ALS?
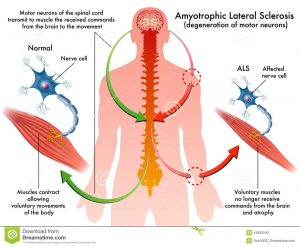
Amyotrophic lateral sclerosis or ALS is a fetal motor neuron disease that brings upon degradation of upper and lower motor neurons. The disease attacks the motor neurons which reach from the brain to the spinal cord and from the spinal cord to the muscles throughout the body. The progressive degeneration of the motor neurons in ALS eventually leads to their demise.
Types of ALS
There are two types of ALS.
Sporadic, which is the most common, affects about 90 to 95 percent of all cases and anyone might get at any time.
Familial, which accounts for 5 to 10 percent of all cases. Familial is inheritable which means a certain mutated gene may be passed on within family members, in those families, there is a 50% chance each offspring will inherit the gene mutation and may develop the disease.
Signs and symptoms of ALS
In the beginning symptoms and signs might be overlooked, they include.
- muscle weakness or muscle atrophy (weakness in the arms and legs first)
- trouble swallowing or breathing
- cramping
As the disease progress the signs and symptoms become severe and widely noticeable. These may vary from person to person.
- Lose in mobility, speech and swallowing
- Cognitive declaim
Sensory nerves and the autonomic nervous system are generally unaffected, meaning the majority of people with ALS maintain hearing, sight, touch, smell, and taste.
What causes ALS?
Scientists have yet to pinpoint what exactly causes the disease but recent research has provided some understanding of what might be going on. A recent study has shown that oxidative stress, mitochondrial damage and protein aggregation might be the problem.
- Oxidative stress and mitochondrial damage cause RNA dysmetabolism
- RNA dysmetabolism causes oxidative stress and mitochondrial damage
- Accumulation of misfolded proteins, which can lead to the imbalance of the protein degradation pathway.
- Iron homeostasis, high levels of iron lead to oxidative stress increase.
Is there a cure?
There is no known cure for ALS but certain studies have shown that the drug Riluzole a glutamate blocker slows the progression of ALS in some people. Also therapy in improving breathing and swallowing has been shown to improve daily living conditions of people with ALS. In addition, people with ALS may experience a better quality of life in living with the disease by participating in support groups and attending an ALS Association Certified Treatment Center of Excellence or a Recognized Treatment Center.
https://www.youtube.com/watch?v=JNZcC_3YLuo
for more information please visit the ALS Association website at http://www.alsa.org/about-als/what-is-als.html?referrer=https://www.google.com/